Patient enrolment has been initiated in a post-market registry for the Combo dual-therapy stent to evaluate its long-term safety and performance in routine clinical practice. The prospective, multicentre, all-comers REMEDEE registry is expected to enrol 1,000 patients.
Researchers at the University of Reading, UK, have been awarded funding of £114,500 by Heart Research UK to investigate new ways of treating heart failure and factors affecting cell division, survival and death.
Gore has announced a favourable ruling involving the Gore Helex Septal Occluder. Judge Joan Ericksen of the US District Court for the District of Minnesota, USA, ruled on 23 July in Gore's favour that the Gore Helex Septal Occluder does not infringe the asserted claims of AGA Medical US patent and granted Gore's request for summary judgement of non-infringement.
Former US president George W Bush's recent heart stent operation will further increase the popularity of what is often an unnecessary and wasteful procedure, says an analyst with research and consulting firm GlobalData.
Symetis has announced the first-time implantation of its ACURATE TA and ACURATE TF transcatheter aortic valve implantation (TAVI) devices in Japan.
Timothy C Ball, an interventional cardiologist at Carilion Roanoke Memorial Hospital, Roanoke, USA, speaks about using a hybrid operating room and explains a transcatheter aortic valve implantation (TAVI) procedure.
CID has announced that it has received the CE mark for longer lengths 31mm in small sizes and 38mm for the entire product range of its polymer-free, drug-eluting stent, Cre8.
The first prospective pilot study of renal denervation in patients with moderate treatment-resistant hypertension indicates that the intervention is safe and effective in this patient population.
A new study indicates that while both access site bleeding and non-access site bleeding after PCI are independently associated with an increased risk of one-year mortality, non-access site bleeding is a stronger correlate of mortality than access site bleeding
Boston Scientific has announced that it has launched a new imaging coronary imaging catheter, the OptiCross, in Europe and in the USA.
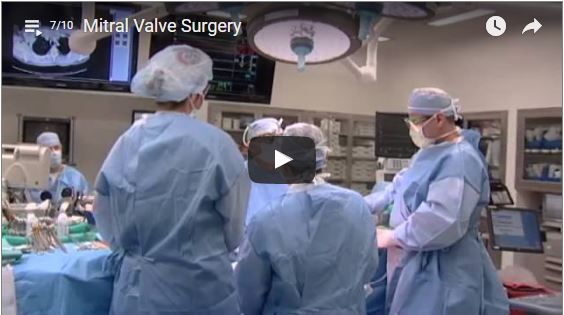
Marc Gillinov and Stephanie Mick, both of the Cleveland Clinic, Cleveland, USA, discuss mitral valve surgery and the options available to patients, including robot-assisted mitral valve repair.
A court in Germany has found that Medtronic's CoreValve infringes Edward Lifesciences' Spenser patent.
Biotronik has launched the Galeo Pro, which is a new range of percutaneous coronary intervention (PCI) guidewires
Corindus Vascular Robotics has announced that for the first time, its CorPath robotic angioplasty device has been used in a PCI procedure.
Sorin has announced that its Vancouver site will manufacture components of the Lotus TAVI valve.
Thoratec Corporation said, in a press release on 1 July, that it has acquired the DuraHeart II ventricular assist system from Terumo for an upfront cash payment of US$13 million and potential future milestone payments, based on regulatory approvals and product sales, of up to US$43.5 million.
Stentys has announced that it has received approval to expand the CE mark indications of its self-apposing coronary stent.
CryoLife announced on 10 July that it has received US Food and Drug Administration (FDA) premarket approval for its new Sologrip minimally invasive laser fibre-optic delivery system.
On the 12 July, Boston Scientific announced it had received US Food and Drug Administration (FDA) 510(k) clearance and CE mark approval for the Guidezilla guide extension catheter and has launched the device in the USA and Europe.
According to an all-comers registry published in EuroIntervention and presented at EuroPCR (21–24 May, Paris, France), cardiogenic shock is the strongest predictor of stent thrombosis following percutaneous coronary intervention.
A small observational study indicates that a self-expanding transcatheter aortic valve implantation (TAVI) device (CoreValve, Medtronic) could potentially be used to treat severe aortic stenosis in patients who have previously undergone mitral valve surgery.
A new NICE guideline aims to improve the outcomes for STEMI patients. The guideline includes recommendations on coronary angiography, primary PCI and reperfusion therapy.
Abbott has announced that the Ministry of Health, Labor and Welfare (MHLW) in Japan has approved the Xience Xpedition everolimus-eluting coronary stent system for the treatment of coronary artery disease.
JenaValve Technology released the first results of its long-term performance and safety in patients with severe aortic stenosis (JUPITER) registry at EuroPCR (21–24 May, Paris, France), demonstrating high procedural success rates and excellent clinical outcomes.
David Holmes (Mayo Clinic, Cardiovascular Diseases and Internal Medicine, Rochester, USA) spoke to Cardiovascular News about the goals of a heart team approach in interventional cardiology
Middle Peak Medical, which is planning to commercialise a novel technology for treating mitral valve disease, has announced that it has US$8.5M in a Series A financing
A study published in the European Journal of Cardio-Thoracic Surgery indicated that 39.6% of patients who received the Revivent Myocardial Anchoring System had a sustained improvement in heart function at one year.
BIBA Medical has announced that it has launched an international TAVI monitor, which is an extension of the company's current TAVI monitor.
Despite white patients and black patients having similar survival rates following aortic valve replacement for aortic stenosis, black patients undergo the surgery significantly less frequently.
StentBoost, a new imaging technique that enhances stent fluoroscopic visibility, is a simple and quick method that offers several advantages during percutaneous coronary intervention in patients with bifurcation lesions
Sachin Goel and Samir Kapadia review the different interventional methods of managing mitral regurgitation
Data from the SEVITENSION study indicate the fixed-dose treatment Sevikar (olmesartan/amlodipine) is superior to perindopril in combination with amlodipine in reducing central blood pressure.
Results from the On-X Heart Valve study presented at the Society for Heart Valve Disease 7th Biennial Congress (22–25 June, Venice, Italy) show that patients on low anticoagulation therapy do well with the On-X prosthetic heart valve.
A new App, for both Mac and Android devices, that provides training on valve-in-valve procedures using transcatheter valves has been launched.
According to several studies presented at the European Society of Cardiology Heart Failure Congress (25–28 May, Lisborn, Portugal), the use of the novel compound Bendavia is associated with improved cardiac function, preserved viable myocardium and increased survival in systolic and diastolic heart failure models.
Medtronic has released the baseline data for the extreme risk patients enrolled in its CoreValve US pivotal trial.
According to data released by Edwards Lifesciences, the Perimount aortic valve is associated with a long-term durability in patients aged 60 or younger at the time of operation.
The joint 2013 European Society of Hypertension/European Society of Cardiology guidelines for the management of arterial hypertension state that lifestyle changes are the "cornerstone" to preventing hypertension.
The FDA has approved Cordis's Adroit 6F guiding catheter for use in various coronary anatomies.
The FDA has given approval to AngioScore to change the instructions for use for its AngioSculpt scoring balloon catheter.
Danny Dvir provides a short summary of practical recommendations that can improve procedural success during valve-in-valve procedures
A new study indicates the cardiac magnetic resonance imaging (MRI) stress testing could be used to predict the risk of cardiovascular death and myocardial infarction in patients with known or suspected coronary artery disease.
A study has found that the transulnar approach for coronary procedures is associated with significantly higher crossover rates compared with the transradial approach.
According to the company the trial will examine the "dissolving" heart device compared to Abbott's drug-eluting stents in patients with coronary artery disease in Japan.
Cardiac surgeons in Germany, Austria and Poland have successfully completed implants and patient enrolment in the VITAL-2 clinical trial for the Vitality Heart Valve System (ValveXchange), according to a company release.
An Oxford-based (UK) master class is available for trainee surgeons to receive training from experience cardiothroacic surgeons and share their own experiences. The class is funded by the Heart UK.
Results from the BIOSOLVE-I study that were presented at the EuroPCR have demonstrated the excellent safety profile of the DREAMS drug-eluting absorbable metal scaffold, according to a company release.
SunTech Medical, in a press release, announced they will be showcasing their ambulatory blood pressure monitoring system for 24 hour management and analysis of patient blood pressure data.
Toshiba has announced that its Infinix DP-i X-ray system is now compatible with its CAT-880B hybrid table for hybrid and transcatheter aortic valve implantation (TAVI) procedures.
According to two European postmarket studies, Abbott Vascular's MitraClip is associated with positive outcomes in patients with mitral regurgitation.
In a study presented at the European Atherosclerosis Society annual meeting (2-5 June, Lyon, France), the first-in-class lipoprotein lipase activator LP071 was shown to reduce plasma levels of triglycerides by almost 100% and more than doubled plasma levels of high-density lipoprotein cholesterol in an animal model.
Biosensors has entered into a licensing agreement with Eurocor on the company’s drug-eluting balloons. The first part of the agreement states that Bionsensors will market and sell drug-eluting balloons manufactured by Eurocor under its own brand.
During a late-breaking trial session at EuroPCR (21–24 May, Paris, France), principal investigator Francesco Prati presented the results of the Demonstr8 study, which showed the Cre8 drug-eluting stent to be non-inferior to an Abbott Vascular bare metal stent with a high statistically significant difference (RUTTS Score <30%; 99.75% Cre8 vs.99.55% bare metal stent; p<0.0001).
Long-term data from the DIVERGE study, presented at EuroPCR (21–24 May, Paris, France), shows that the Axxess drug-eluting stent is associated with safe and effective treatment of complex coronary lesions.
Maquet has announced it has acquired the company LAAx and its TigerPaw system II occlusion device.
Registry data presented at EuroPCR (21–24 May, Paris, France) shows the Neovasc Reducer, a novel percutaneous device that is CE marked for the treatment of refractory angina, to improve cardiovascular parameters in refractory angina patients.
After receiving the CE mark for its dual therapy Combo stent, OrbusNeich has launched the device onto to the European market.
A late-breaking study, ADVISE (Adenosine vasodilator independent stenosis evaluation) II, presented at EuroPCR (21–24 May, Paris, France) indicates that the combination of instant wave-free ratio (iFR) and fractional flow reserve (FFR) is a useful clinical approach for assessing coronary lesions.
Kona Medical presented three- and six-month data from its WAVE I study of its Surround Sound renal denervation system in a late-breaking trial session at EuroPCR (21-24 May, Paris, France).
Results from a large observational study, presented at EuroPCR (21–24 May, Paris, France) suggest that in the absence of GP IIb/IIIa receptor inhibitors, bivalirudin does not improve outcome compared with heparin in patients with non-ST segment elevation acute coronary syndrome undergoing percutaneous coronary intervention.
A late-breaking trial, presented at EuroPCR (21–24 May, Paris, France), indicates that Biotronik's Orsiro a drug-eluting stent with a biodegradable polymer was non-inferior to Abbott Vascular's Xience Prime, a drug-eluting stent with a durable polymer, in terms of in-stent late lumen loss.
Daniel Steinberg reviews the transradial approach in primary percutaneous coronary intervention (PCI) and looks at why the USA has been reluctant to adopt the approach despite it reducing access site and bleeding complications compared with the transfemoral approach
Renu Virmani, medical director, CVPath Institute, Gaithersburg, USA, spoke to Cardiovascular News about bioresorbable stents and their potential impact on the future management of coronary artery disease.
Morton Kern is chief of cardiology at the Long Beach Veterans Administration Hospital, Long Beach, USA, and is also associate chief of Cardiology at University of California, Irvine, Orange California, USA. He is the co-chair of the Society for Cardiovascular Angiography and Interventions' annual meeting (SCAI; 8–11 May 2013, Orlando, Florida). He talked to Cardiovascular News about his career and his achievements
Interim data from the REDUCE-HTN study, which was presented at EuroPCR (21–24 May, Paris, France), indicates that the Vessix renal denervation system (Boston Scientific) is associated with a significant and sustained reduction in blood pressure.
Presented at EuroPCR, the one-year results from BELLO study showed durability following treatment of de novo lesions with the novel angioplasty device.
CardioKinetix has announced positive clinical data that showed positive results for patients treated with minimally invasive structural heart device for heart failure.
At EuroPCR 2013 in Paris, France (21–24 May), three separate analyses have addressed one of the most common clinical challenges in the treatment of complex coronary artery disease and showed that the the Resolute drug-eluting stent from Medtronic has performed strongly in coronary bifurcation lesions over the long term.
JenaValve Technology released the first results of its long-term performance and safety in
patients with severe aortic stenosis (JUPITER) registry at EuroPCR (21–24 May, Paris, France), demonstrating high procedural success rates and excellent clinical outcomes.
Direct Flow Medical has announced that it met its primary endpoint in the fully-enrolled, 100-patient DISCOVER CE mark trial by achieving 99% freedom from all-cause mortality at 30 days.
According to a press release clinical evidence supporting the benefits of using the MGuard EPS in STEMI patients continues to grow and a presentation at EuroPCR meeting added important new data underlining the protective benefits of MGuard six months post procedure.
According to a presentation at EuroPCR, "real world" ADVANCE study has confirmed that using CoreValve has good economic value in patients unable to undergo surgery.
According to Abbott, Xience Xpedition is the world's longest coronary drug-eluting stent and has received the CE mark.
Of those studied, only 4.3% were reported as experiencing a primary endpoint event in the e-BioMatrix study.
The ORBIT II trial evaluating safety and effectiveness of the orbital atherectomy system in treating calcified coronary lesions is expected to presented as late-breaking data at EuroPCR (21–24 May, Paris, France).
One-year results from SOURCE XT, one of the largest, post-approval transcatheter aortic valve implantation (TAVI) registries (according to a EuroPCR press release) has reported good clinical outcomes in clinical practice.
The DESolve Nx trial treated target lesions in 126 patients with single de novo coronary artery lesions with the DESolve device. Results showed the primary endpoint of in-stent late lumen loss was 0.21mm (+0.34) at six months.
According to a Medtronic press release, data from the global SYMPLICITY registry showed renal denervation with the Symplicity system met performance expectations in a real-world setting.
According to an online survey of 141 interventional cardiologists worldwide, the use of drug-eluting stents and the delay to healing is a concern.
Philips announced that their CX50 xMATRIX now has 2D intracardiac echo capability. The CX50 xMATRIX with available Live 3D TEE and ICE is expected to be shown in Paris, France, at EuroPCR, 21–24 May.
According to a clinical study and Philips, using the AlluraClarity live guidance system, a 50% reduction in X-ray dose could be achieved without any loss in image quality.
Data of the randomised JACK-EPC trial showed that the Genous stent had a significantly lower rate of binary restenosis compared to bare metal stents in acute coronary syndrome patients. The results have been published in Minerva Cardioangiologica.
Medtronic announced that its Export Advance aspiration catheter recently received the CE mark and will soon be launched in Europe and other international markets.
Published in Circulation, new research examining the frequency and composition of embolic debris captured in TAVI, has found that in a 40 patient series, there were visible debris in 75% of patients when using the Claret Montage Dual Filter System.
A symposiumat EuroPCR (21–24 May, Paris, France) is expected to discussed the Cre8 stent. According to a company release, this will include presentations from clinicians on Cre8 trial data, followed by a panel and audience discussion.
On the 20 May 2013, Biosensors International announced it has received CE mark approval for its BioMatrix NeoFlex, the latest addition to the BioMatrix family of drug-eluting stents.
Circulite has announced it has received CE mark approval to conduct a multicentre trial on the Synergy IC circulatory support system. An abstract on the system is expected to be presented at EuroPCR (21–24 May, Paris, France).
A new study suggests that transcatheter aortic valve implantation (TAVI) devices are under used in Europe, and that this under use is associated with economic status and reimbursement strategies.
On-X Life Technologies has announced that it has completed enrolment for low-risk aortic valve and mitral valve patient groups in its Prospective randomised On-X Valve anticoagulation trial (PROACT).
Elixir Medical has announced it has received Food and Drug Administration (FDA) approval to start its EXCELLA clinical trial of its Desyne Nx novolimus-eluting stent system.
Sorin has announced that it has received conditional approval from the Food and Drug Administration (FDA) to conduct an investigational device exemption (IDE) study of its Perceval S Sutureless aortic valve.
A new risk score, developed using data from the RISK-PCI study, could be used to identify which patients are at high risk of bleeding after undergoing a percutaneous coronary intervention (PCI) procedure.
A study published in Circulation indicates that compared with white patients, black and Hispanic patients have worse outcomes following percutaneous coronary intervention (PCI).
According to a study presented at EuroPRevent 2013, mental vulnerability is associated with a significantly increased risk of fatal and non-fatal cardiovascular disease independently of classical risk factors.
The REMEDEE study, which showed that OrbusNeich's Combo dual therapy stent is effective at controlling neo-intimal proliferation, has been published in the journal JACC: Cardiovascular Interventions.
Symetis has announced that a post-market study has confirmed that its transcatheter aortic valve implantation (TAVI) Acurate TA device is associated with a high procedural success rate.
Results from the C-CURE study, which reviews the use of C3BS-CQR-1 in patients with chronic heart failure, have been published in the Journal of the American College of Cardiology.
Maquet has received both FDA approval and CE mark approval for its AIR-BAND radial compression device.
In March, Natec Medical received a licence to market its Tamarin Blue and Filao RX in Canada
MediLive has developed a 3D training series for both students and medical professionals.
Max Baghai and Olaf Wendler review the current data for TAVI procedures and discuss how the complications of these procedures can be minimised.
A study published ahead of print in the Journal of the American College of Cardiology indicates that following implantation of a left ventricular assist device, sexual function does not improve and may even worsen.
A new study, published ahead of print in the Journal of the American College of Cardiology, indicates that renal denervation delivered through extracorpeal high-intensity focused ultrasound may be an alternative approach to managing resistant hypertension.
BioControl Medical has announced that the FDA has approved full expansion of the INNOVATE-HF study, which is investigating BioControl Medical's CardioFit device for vagal nerve stimulation.
Maquet Medical Systems has announced that it has launched the MIRA-iCS Retractor system, for use during minimally invasive revascularisation procedures, on to the USA market.
Arthesys has announced that it has received the CE mark for its fourth generation Maya coronary balloon catheter.
Bayer has announced that the European Medicines Agency has recommended that its drug rivaroxaban (Xarelto) be approved for the prevention of atherothrombotic events in patients with acute coronary syndromes (ACS).
NeuroVive has announced the 600th patient has been recruited to its pivotal Phase III European, multicentre trial (CIRCUS trial), which is assessing CicloMulsion.
Boston Scientific has announced that the Platinum China study, which was presented at China Interventional Therapeutics (20–23 March, Beijing, China), indicated that the Promus Element stent is associated with a low rate of stent thrombosis at nine months.
The UK's National Institute for Health and Clinical Excellence (NICE) has launched a new quality standard for hypertension, which states that person-centred care is vital in the management of the condition.
The FDA Circulatory System Devices Panel of the Medical Devices Advisory Committee voted five to three that the benefits of Abbott Vascular's MitraClip outweigh its risks.
A new study, published ahead-of-print in the Journal of Thoracic and Cardiovascular Surgery, suggests that the Trifecta aortic valve has a similar haemodynamic performance to that of a healthy aortic valve.
For the first time, the Promus Premier everolimus-eluting coronary stent has been implanted into patients in Ireland and in the Netherlands.
At ACC, data from the ORBIT II study showed that patients treated with the Orbital atherectomy system (Cardiovascular Systems) were associated with an 89.8% freedom from major adverse cardiovascular events rate.
According to the company, the Aquilion ONE ViSION can image the heart in one rotation with a low dose of radiation when used in conjuction with the company's Vital Images' CT Myocardial Analysis software. A trial conducted by the National Institutes of Health confirms in 107 patients that the system's average radiation dose was low.
At the American College of Cardiology Scientific Session (ACC.13), Philips introduced its portfolio of cardiology solutions. This included the IntelliSpace ECG, the ST80i Stress Testing System, the EchoNavigator, and the Corindus CorPath 200 System.
Despite a range of safe and effective antihypertensive therapies, hypertension is still a challenging condition to control only a small percentage of hypertensive patients achieve adequate blood pressure control. Intravascular renal denervation may be beneficial in treating resistant hypertension, but there are gaps in the evidence base. Jan Staessen reviews the current research for this intervention and the priorities for future research
The midtem results from cohort A of the PARTNER study indicate that, after three years, the Sapien transcatheter aortic valve is still structurally intact and that transcatheter aortic valve implantation continues to be comparable to surgery in high-risk patients.
New study, CHAMPION PHOENIX, indicates that cangrelor significantly reduces the rate of ischaemic events including stent thrombosis compared with clopidogrel in patients undergoing urgent or elective percutaneous coronary intervention.
Two-year data for the Symplicity renal denervation system showed that the intervention was associated with a sustained significant drop in blood pressure.
One-year data from APPOSITION III, which is assessing the long-term performance of the Stentys Self-Apposing stent, indicated that the device is associated with a very low rate of mortality (2%).
Results presented at the ACC for the bioresorbable vascular scaffold Absorb showed that the device was associated with a major adverse cardiovascular events rate of 10% at three years.
Results of the DISCOVER trial, presented at ACC 2013, have shown that patients treated with the Direct Flow Medical Transcatheter Aortic Valve System (Direct Flow Medical) achieved excellent survivability and sustained haemodynamic improvements with minimal occurrence of aortic regurgitation at six months.
Results from the IMPACT trial, presented at ACC 2013, have found that cardiologists changed their diagnostic testing strategy of coronary artery disease in 60% of female patients following Corus CAD testing.
At the ACC 2013 sessions, Ian Meredith, Melbourne, Australia, reported clinical endpoint data from the PLATINUM workhorse clinical trial comparing the safety and effectiveness of the Promus Element Coronary Stent System (Boston Scientific) to the Xience V Everolimus-Eluting Coronary Stent System (Abbott).
Data from the Global RESOLUTE clinical programme indicates that interrupting dual antiplatelet therapy one month after a patient is implanted with a Resolute drug-eluting stent is not associated with an increased safety risk.
New data for the MitraClip system indicates that the device is associated with a significantly lower rate of 30-day mortality compared with open surgery.
The study is aimed to evaluate an enhanced version of the Carillon Mitral Contour System (Cardiac Dimensions), a minimally-invasive therapy designed to treat heart failure patients suffering from functional mitral regurgitation.
The Ilumien Optis system combines fractional flow reverse and intravascular optical coherence technology (OCT) to help with stent placement and has been launched by St Jude Medical in Japan.
Siemens Healthcare has announced that the FDA has approved its Artis Q and Artis Q.zen angiography system families.
CircuLite has announced it has received conditional FDA approval for an investigational device exemption study of its Synergy circulation support system.
Rox Medical has announced that the first patients in Ireland have been enrolled in the in CONTROL-HTN hypertension at Beaumont and Mater private hospitals.
Royal Philips Electronics has announced that it has received 510(k) clearance from the FDA to market its EchoNavigator live image-guidance tool.
Medtronic has been given the go-ahead to update the dual antiplatelet therapy CE mark labelling for its Resolute Integrity drug-eluting stent.
The FDA has approved two Humanitarian Use Device (HUD) designations for the new SynCardia's 50cc total artificial heart
Medtronic has announced it has received FDA approval to conduct an early feasibility study of its native outflow tract transcatheter pulmonary valve.
Neovasc has announced that the clinical protocol for its COSIRA study, which assesses the company's novel percutaneous device for reducing refractory angina, has been published in a peer-reviewed journal.
Medtronic has received the CE mark for its Engager transcatheter aortic valve implantation system, which delivers the Engager valve transapically.
Stefan D Anker is the 2012–14 president of the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). He has been credited with establishing cachexia as an independent prognostic factor for cardiovascular illness, and he has shown that iron deficiency is a valid therapeutic target in heart failure. He spoke to Cardiovascular News about his career, research in heart failure and his goals during his HFA presidency tenure.
Stefano Savonitto, director, Cardiology Division, Arcispedale S Maria Nuova, Reggio Emilia, Italy, talked to Cardiovascular News about treating older patients with STEMI. He spoke about this topic at JIM.
According to a new study, transcatheter aortic valve implantation is feasible in non-surgical patients with severe native aortic valve regurgitation without stenosis.
Cardiovascular Systems presented three-year data for its orbital atherectomy system at CRT 2013 (23–26 February 2013, Washington, USA).
A new study indicates that microvascular resistance, contrary to previous studies, increases after percutaneous coronary intervention and this increase is not linked to increasing epicardial stenosis.
The FDA has approved the 34mm and 38mm lengths of Medtronic's Resolute Integrity drug-eluting coronary stent.
Direct Flow Medical has been awarded two patents, one from the US Patent and Trademark Office and one from the Japanese Patent Office, for its transcatheter aortic valve implantation system.
According to a study published in Circulation: Cardiovascular Genetics, the Corus CAD test outperforms myocardial perfusion imaging in overall diagnostic accuracy for obstructive coronary artery disease.
St Jude Medical has announced it has initiated a study to evaluate whether the renal denervation can lower the risk of major cardiovascular events as well as lower blood pressure
The IDE study is designed to explore alternative access routes with transcatheter aortic valve implantation (TAVI). It is believed to be a first for any medical specialty society.
ReCor Medical has announced that it has begun ACHIEVE a post-marketing study of its next-generation, ultrasound-based renal denervation system, Paradise.
Intracoronary administration of progenitor cells, in patients with acute myocardial infarction or patients with chronic heart failure, has an adequate safety profile
The Resistant Hypertension Course, a joint initiative of the European Society of Hypertension (ESH), the European Association of Percutaneous Cardiovascular Interventions (EAPCI) and PCR, will take place this week.
José M Pérez-Pomares, member of the European Society of Cardiology (ESC) Working Group on Development, Anatomy and Pathology, reviews the effects of ischemic heart disease and discusses the biological foundations of cell-based approaches to repair the damaged (infarcted) heart.
Despite the perception that financial conflicts of interest influence the outcome of trials, a new study indicates that self-reported financial conflicts of interests are not associated with an increased likelihood of favourable results.
Cardiorentis has initiated the first-ever acute heart failure (AHF) Phase III trial to be specifically designed to assess the effect of early treatment on cardiovascular mortality.
Information about OrbusNeich's Combo dual therapy stent will be presented during the Joint Interventional Meeting (JIM; 14–16 February, Rome, Italy)
According to a company release, the Promus Premier (Boston Scientific) is designed to provide physicians improved drug-eluting stent performance in treating patients with coronary artery disease.
The UK's National Institute for Health and Clinical Excellence (NICE) has issued a draft version of its guideline for acute management of ST-segment-elevation myocardial infarction (STEMI) for public consultation
The first patient in Svelte Medical Systems's DIRECT II (Direct Implantation of Rapamycin-Eluting stents with bioabsorbable drug Carrier Technology) study, the company announced, has been treated.
Therox has announced it begun an investigational device exemption study of its next generation super saturated oxygen therapy for patients with acute myocardial infarction.
St Jude Medical has announced that the first patient in its study evaluating the 25mm Portico transcatheter heart valve has been implanted with the device.
Kips Bay Medical has announced that the first patient in its US clinical feasibility trial has been implanted with the eSVS Mesh graft.
According to Natec Medical, the Tamarin Blue PTCA RX dilatation catheter features exceptional trackability and pushability, and a unique crossability into the tight stenosis with its tapered tip.
Philippe Pibarot reviews the use of stress imaging to determine the severity of aortic stenosis and its management. He spoke about this topic at PCR London Valves (30 September–2 October 2012, London, UK).
A new study indicates that post dilation, following balloon expandable TAVI, is a safe and effective way to reduce paravalvular regurgitation
This post-market clinical study will further evaluate the safety and efficacy of the EnligHTN Renal Denervation System (St Jude Medical) in patients with uncontrolled hypertension.
Survival benefit found with surgical ventricular reconstruction plus coronary artery bypass graft procedure
The CE mark approval for BioFreedom, a polymer-free drug-coated stent, was supported by strong data from the BioFreedom first-in-man study.
According to a press release, the Direct Flow Medical Transcatheter Aortic Valve System is designed to virtually eliminate aortic regurgitation by allowing complete assessment of haemodynamic performance, repositioning and retrieval after the valve is fully deployed in the native valve annulus.
A team at Peter Munk Cardiac Centre, University Health Network, Toronto, Canada,...
According to a company release, the treatment parameters of this new device have been further enhanced to reduce energy delivery to 30 seconds by maximising cooling of the endothelium and efficiently treating the nerves circumferentially.
The objective of this study is to demonstrate the initial safety and performance of the eSVS Mesh for use as an external saphenous vein graft support device during coronary artery bypass surgery.
The company announced that this new code will provide additional reimbursement for the enCorSQ, a new approach to the long-term treatment of mitral valve disease.
Conventional thinking has been that CPR is futile after 20 minutes, but researchers in the USA have found that their study results challenge that assumption.
OrbusNeich is holding a symposium about its COMBO stent at AsiaPCR/SingLIVE 2013 (24–26 January, Singapore)
Placed percutaneously, a company press release explains, OneShot delivers radiofrequency energy in a circumferential manner to the renal arterial wall, and requires only a single treatment per artery.
The HeartMate II Risk Score, as outlined in the Journal of the American College of Cardiology, could be used to estimate mortality in patients receiving a HeartMate II left ventricular assist device
According to a study published in Catheterization and Cardiovascular Interventions, ambulatory percutaneous coronary intervention in patients with stable coronary artery disease is safe and effective
CardioLogical Solutions has been given an additional patent for its aortic embolic protection devices
Phoenix Cardiac Devices states a new study will assess the safety and efficacy of its BACE device for the treatment of mitral regurgitation with the goal of obtaining CE Mark approval
The Pharmacovigilance Risk Assessment Committee of the European Medicines Agency has recommended that the marketing, supply and authorisations for the laropiprant/nicotinic acid containing drugs Tredaptive, Pelzont and Trevaclyn be suspended
A 39-year-old female with New York Heart Association (NYHA) class III heart failure has become the first patient to be implanted with the CardioKinetix's Parachute device in the company multicentre PARACHUTE IV trial
Following on from the use of percutaneous technology in peripheral vascular disease and in coronary artery disease, percutaneous technology is now being used to treat valvular dysfunction. Jose L Navia and Sharif Al-Ruzzeh review its application in this context
As well as receiving the CE mark for its DS1000 mitral valve repair device, NeoChord has also started a 50-patient registry to assess the first European procedures with the device
Abbott's next-generation Xience Xpedition drug-eluting stent system has received FDA approval and has been launched onto the US market
ACES is a randomised study designed to demonstrate the positive clinical benefit and impact on resource utilisation of the Svelte Acrobat Integrated Delivery System (IDS) compared with conventional stent devices.
The TRIS (TandemHeart to Reduce Infarct Size) trial will evaluate the effectiveness of ventricular unloading on the reduction of infarct size for patients who have suffered a severe heart attack.
Study aims to improve clinical outcomes by offering physicians more information to further understand how FFR and OCT influence physician decision-making for coronary intervention
Cosmo Godino, Cardio-Thoracic-Vascular Department, San Raffaele Hospital, Milan, Italy, writes that not all efforts should be directed toward technical and procedural aspects to achieve final chronic total occlusion-PCI recanalisation.
The world's first low profile, 14 French pre-mounted, pre-crimped, and pre-packaged, ready-for-use, TAVI system reduces time from sterile package to valve deployment.
Published data demonstrated that the Symplicity renal denervation system provides blood pressure reduction in patients with treatment-resistant hypertension sustained to 12 months.
The BIOHELIX-I trial evaluates the safety and performance of the PRO-Kinetic Energy coronary, bare metal stent.
Biosensors has announced the enrolment of the first patient in the LEADERS FREE study involving BioFreedom, the polymer-free drug-coated stent.
Celladon has announced that it has dosed the first European patient in its ongoing international Phase 2b clinical trial of MYDICAR.
Matthew Gillespie, Philadelphia, USA, and others have found that transcatheter pulmonary valve implantation with the Melody valve (Medtronic) may be an effective treatment option for patients with failed bioprosthetic valves.
A study published ahead of print in the Journal of the American College of Cardiology indicates that the rates of readmission following axial flow left ventricular assist device (LVAD) implantation decrease during the first six months and thereafter stabilise.
Long-term studies (21 years) and 30 years of clinical use of the Mitroflow valve have demonstrated excellent haemodynamic performance, great durability and ease of use.
Revivent (BioVentrix) offers an alternative less-invasive option than surgical ventricular restoration to repair the left ventricle from damage done by a heart attack, enhancing performance of the heart's non-damaged myocardium.
EVOLVE II is designed to further assess the safety and effectiveness of the Synergy Stent System (Boston Scientific) and support FDA and Japanese regulatory approvals for the treatment of atherosclerotic coronary lesions.
The new survey was undertaken by the American Medical Group Foundation, and released at the launch of the US healthcare campaign "Measure Up, Pressure Down".
ORBIT II is evaluating the safety and effectiveness of Cardiovascular Systems' atherectomy technology in treating severely calcified coronary arteries.
The funding will be used to further validate the company's transcatheter mitral valve implantation technology and is expected to carry the company through its feasibility and CE mark clinical trials.
Results from the BELLO study indicate that the use of a paclitaxel-eluting balloon for the treatment of de novo small vessel disease is associated with less angiographic late loss and similar rates of restenosis and revascularisation compared with a paclitaxel-eluting stent.
Thomas Pilgrim, Bern, Switzerland, and others reported in Circulation: Cardiovascular Interventions that intercurrent events, such as access and bleeding complications, increase the cumulative risk of stroke and death after TAVI.
The PROMISE study will test comparative effectiveness of anatomical versus functional non-invasive diagnostic tests for the assessment of stable symptomatic patients with possible coronary artery disease.
The Journal of the American College of Cardiology (JACC): Heart Failure will begin publishing in February 2013 on a bimonthly basis.
For the first time, the National Cardiovascular Data Registry (NCDR) has published in the Journal of the American College of Cardiology a general report of data from the CathPCI registry, which characterises the clinical profile and outcomes of patients undergoing cardiac catheterisation and percutaneous coronary intervention procedures. Lead author of the report, Gregory Dehmer, reviews the report's key findings.
NICE has recommended ivabradine as an option for the treatment of people with chronic heart failure (NYHA class II to IV) with systolic dysfunction, who are in sinus rhythm and whose heart rate is 75 beats per minute or more and who have a left ventricular ejection fraction of 35% or less.
The Tryton pivotal study is an international randomised trial that compares a Tryton stent in the side branch vs. conventional provisional stenting in the side branch, with both arms of the trial utilising a standard drug-eluting stent in the main vessel.
According to a company release, in both the Artis Q and Artis Q.zen series, the new X-ray tube can help to identify small vessels up to 70% better than conventional X-ray tube technology.
The transapical procedure was performed by Anson Cheung, associate professor of surgery and director of cardiac transplant at St Paul's Hospital in Vancouver, Canada.
Sync-Rx develops advanced software applications that optimise and facilitate transcatheter cardiovascular interventions using automated online image processing.
Cardio3 BioSciences has announced it has received authorisation from the Belgian Federal Agency for Medicines and Health Products (FAMHP) to begin its CHART-1 (Congestive heart failure cardiopoietic regenerative therapy) European phase III trial for C3BS-CQR-1, an autologous stem cell therapy for heart failure, in Belgium.
David Holmes, past president of the American College of Cardiology, principal investigator of the PROTECT AF trial and other several other studies in electrophysiology and interventional cardiology, spoke to Cardiovascular News about his career, mentors, research and interests outside medicine.
The approval was supported with data from the ADVANCE study, in which patients who received the HeatWare device achieved a 94% survival at six months. The trial also met its primary endpoint of establishing non-inferiority between the investigational device and comparator arm of the study.
Corus CAD (CardioDX) is a decision-making tool that can help physicians exclude obstructive coronary arterial disease as the cause of a stable non-diabetic patient's symptoms.
The valve that can be completely resheathed, repositioned or retrieved is designed for patients with severe aortic stenosis who are considered to be inoperable or high risk for conventional open-heart valve replacement surgery.
"the Gore REDUCE clinical study shares notable similarities to the RESPECT study which we feel will help lead to a beneficial study outcome," said Gore REDUCE principal investigators.
The SURTAVI trial will evaluate whether the CoreValve system (Medtronic) is non-inferior to surgical aortic valve replacement, based on the composite primary endpoint of all-cause mortality and disabling stroke at 24 months.
The Federal Circuit Court of Appeals has affirmed the April 2010 jury verdict from the Federal District Court of Delaware that the CoreValve System infringed a single Andersen patent held by Edwards Lifesciences.
Gregory W Stone, New York, USA, presented one-year results, at TCT, from a substudy of the largest study of its kind the ADAPT-DES registy showing that stent procedures guided by intravascular ultrasound (IVUS) resulted in better patient outcomes and fewer complications at 30 days and 12 months compared to procedures without IVUS, and were safe.
This catheter is specifically designed to facilitate quick and accurate thrombus aspiration in myocardial infarction patients with ST-segment elevation.
Improvements in haemodynamics and quality of life were demonstrated at 24 months in an extended analysis of the pilot study which assessed vagus nerve stimulation therapy for heart failure patients with the CardioFit System (Biocontrol Medical).
Reporting on the results of an economic analysis of the FAME II study, at TCT, William Fearon, Stanford, USA, said that fractional flow reserve (FFR) guided percutaneous coronary intervention (PCI) was cost-effective, compared with medical management, in patients with stable coronary disease (CAD).
In the 86.3% of non–ST-elevation acute coronary syndrome (NSTE-ACS) patients with elevated hs-TnT, ticagrelor (Brillinta, Aztrazeneca) tablets reduced the composite of cardiovascular death, myocardial infarction, and stroke.
Results showed that ticagrelor plus aspirin, compared to clopidogrel plus aspirin, reduced cardiovascular death, myocardial infraction and stroke in acute coronary syndrome patients regardless of coronary artery disease extent and treatment strategy.
The objective of this study is to demonstrate the initial safety and performance of the eSVS Mesh for use as an external saphenous vein graft (SVG) support device during CABG procedures.
Vessix Vascular is the developer of the Vessix Vascular V2 Renal Denervation System which has received CE mark in Europe and TGA approval in Australia.
Jay H Traverse, Minneapolis Heart Institute at Abbott Northwestern Hospital, Minneapolis, USA, and colleagues have found that timing of cardiovascular cell delivery within the first week after a heart attack does not have any effect improving left ventricular function following reperfusion.
The new heart device is used for simultaneous implantation of an aortic tissue valve and ascending aorta. It allows for safer device manipulation during assembly and implantation, thus limiting cardiac surgery time.
The study met its primary endpoint showing the MGuard Embolic Protection Coronary Stent was significantly superior to the control arm of bare metal and drug-eluting stents in the treatment of heart attack patients.
Joshua M Hare, Miami, USA, and colleagues have found that "transendocardial injection of allogeneic and autologous mesenchymal stem cells were both associated with low rates of treatment-emergent serious adverse events, including immunologic reactions in patients with left ventricular dysfunction due to ischaemic cardiomyopathy.
The move strengthens Medistim's commitment to the UK market following NICE recommendation of its VeriQ flow measurement system for assessing graft blood flow during coronary artery bypass graft surgery.
A comparative study has shown, at five years, that diabetic patients who underwent bilateral internal thoracic artery grafting had a 13% greater survival rate, as well as a lower risk for non-fatal heart attacks, recurrent chest pain, and future re-interventions, compared to patients treated with percutaneous coronary intervention.
Synergy (Boston Scientific) features a PLGA polymer and everolimus drug coating that dissipates by three months.
A large comparative registry undertaken in a "real-world" setting has shown that, at five years, coronary artery bypass grafting demonstrated lower rates of death, myocardial infarction and target vessel revascularisation compared to percutaneous coronary intervention in patients with left main coronary artery or multivessel disease. Results were presented at EACTS.
First 30-day results of the Engager European pivotal trial support the safety and performance of the Engager transapical aortic valve system (Medtronic) in patients with severe aortic valve stenosis at high risk for surgical aortic valve replacement.
Non compliant percutaneous transluminal coronary angioplasty (PTCA) balloons are commonly used to post-dilatate stents and to treat in stent restenosis.
The Biotronik Pantera Lux Paclitaxel Releasing Balloon is a novel treatment for restenotic coronary artery lesions after drug-eluting or bare metal stenting.
This new fibre-optic device will allow clinicians to provide a higher-efficacy, faster intra-aortic balloon counterpulsation therapy to patients 5'0" to 5'4" tall.
Data presented at TCT show that the primary analysis was not statistically significant but trended towards superiority with the Amplatzer device, while additional analyses demonstrated superiority.
Target lesion revascularisation rates at three years were significantly lower in favour of the DESyne stent as compared to the Endeavor zotarolimus-eluting stent (1.4% vs. 9.9%; p=0.008).
The VidaEcho system features a simulated 3D anatomy of the heart, and the corresponding echo image; by moving a virtual echo probe handle with a mouse, users learn how the device will interact with the cardiac anatomy.
MGuard Embolic Protection Stent (InspireMD) shows a significant 29% improvement in complete ST resolution compared to bare metal or drug eluting stents.
Contrary to previous data, STEMI patients with prior open heart surgery, or coronary artery bypass graft surgery (CABG) and receive a coronary stent have similar outcomes to patients without previous CABG, based on study of a large, prospective, regional STEMI network, presented at TCT.
The Revivent system is intended for patients suffering from ischaemic cardiomyopathy.
This language outlines a minimum duration of three months of DAPT for certain patients who may need to interrupt or discontinue the medication for a variety of reasons and supports the strong safety profile for Promus Element and Promus Element Plus stents.
A semi-structured survey suggests that more than 15% of patients suffering from severe symptomatic aortic stenosis eligible for transcatheter aortic valve implantation (TAVI) did not receive the treatment for budget-related reasons in 2011.
After three years of follow-up, the benefits of TAVI were sustained as measured by all-cause mortality, cardiovascular mortality, repeat hospitalisation, and functional status, according to new data presented at TCT.
The ISAR-LEFT MAIN 2 trial is the first and largest performed randomised, multicentre comparison trial between zotarolimus-eluting and everolimus-eluting stents in unprotected left main coronary artery lesions.
"This analysis adds provocative new insight to ongoing discussions about the minimal duration of dual antiplatelet therapy required to safeguard patients with the latest generation drug-eluting stents," said Sigmund Silber, presenter of the RESOLUTE data at TCT.
Under this FDA-approved IDE, up to 880 ST-elevation myocardial infarction (STEMI) patients at 50 sites in the USA and worldwide will be enrolled in the APPOSITION V clinical trial.
The studies evaluated hospital readmission rates for subsequent heart attack and initial hospitalisation costs in patients with acute coronary syndrome treated with a percutaneous coronary intervention and antiplatelet therapy, including prasugrel or clopidogrel.
The PrimeWire Prestige Plus device will be used in six new Volcano-sponsored clinical studies to explore new indications in stable and unstable coronary artery disease, peripheral arterial disease and neurology.
The Kona Surround Sound system utilises focused ultrasound, delivered from outside the body, to treat the nerves leading to and from the kidney.
Riociguat demonstrates a statistically significant improvement in the six-minute walk test, a predictor of improved outcome in patients suffering from pulmonary arterial hypertension.
The primary endpoint, freedom from all-cause mortality from procedure to 30 days, was met at 97%. Freedom from all cause cardiovascular mortality at 30 days was 100%.
BioMatrix Flex significantly reduced the risk of clinical events compared with Cypher Select, which was associated with a reduced risk of very late stent thrombosis.
The Parachute Ventricular Partitioning Device (CardioKinetix), available now in eight sizes, is a minimally invasive treatment used for percutaneous ventricular restoration therapy.
Permaseal is a transapical access device designed to enable self-sealing, sutureless cardiac access and closure of structural heart repair procedures including TAVI and mitral valve repair.
The Platinum Long Lesion study met its primary endpoint of target lesion failure at 12 months with a 3.2% rate for the Promus Element Stent (Boston Scientific) in the per protocol population compared to a pre-specified performance goal of 19.4% based on historical outcomes for the Taxus Express Stent.
Gore is introducing the technology in the REDUCE study for prevention of recurrent stroke in patent foramen ovale patients.
Medtronic has announced new findings from the Medtronic CoreValve ADVANCE study for one year survival and health-related quality of life.
The Resolute drug-eluting stent from Medtronic delivered successful clinical results two challenging characteristics of coronary artery lesions commonly found in patients with diabetes.
Approved by the FDA under an investigational device exemption, the study will evaluate the safety and effectiveness of the device in approximately 370 patients at up to 30 sites in the USA.
New data presented at the TCT congress show sustained blood pressure reduction and safety with Symplicity Renal Denervation system in patients with treatment-resistant hypertension.
An analysis of the benefits to using a fractional flow reserve-guided intervention strategy found that the technology can improve patient outcomes while saving significant amounts of money.
Acute cardiac care experts who met at Acute Cardiac Care Congress 2012 in Istanbul, Turkey, said that patients with severe chest pain should call the emergency number immediately as pre-hospital care is essential to survival.
Data suggest Sidegaurd stent has long term benefits in treating bifurcation disease
The expanded indication includes patients with aortic valve stenosis who are eligible for surgery, but who are at high risk for serious surgical complications or death.
Denise Fischer, Homburg, Germany, says that renal denervation may have a positive effect in psychological processes, stress perception and processing as well as in quality of life.
A patient with a functional bicuspid valve is less likely to require repeat valvuloplasty, aortic valve replacement, death, or a heart transplant than a patient with other types of valve morphology, according to a study published by Shiraz Maskatia, Texas Children's Hospital, Houston, USA, and colleagues in Catheterization and Cardiovascular Interventions.
Akshay Bagai, Toronto, Canada, and colleagues have found that mentored simulation may help less proficient cardiac catheterisation operators to improve their skills.
The US prospective, multicentre, single-arm, clinical study will evaluate the safety and efficacy of the new Gore Septal Occluder in the treatment of percutaneous, trancatheter closure of ostium secundum atrial septal defect.
Cibiem will develop a proprietary, minimally invasive, catheter-based approach focused on carotid body modulation for the treatment of sympathetic nervous system-mediated diseases.
In the survey, conducted by the Society for Cardiovascular Angiography and Interventions (SCAI), four out of five patients say their lives have changed for the better following angioplasty.
The device, which is also CE-marked, features a balloon specifically designed to prevent balloon ruptures in transcatheter aortic valve implantation (TAVI) and balloon aortic valvuloplasty (BAV) procedures.
The report published online in The Journal of the American College of Cardiology (JACC) analyses data from 1.1 million patients who underwent diagnostic cardiac catheterisation procedures and 941,248 patients treated with percutaneous coronary intervention from January 2010 to June 2011.
The collaboration is focused on integrating Infraredx's true vessel characterisation (TVC) Imaging System with Philips' Allura Xper catheterisation (cath) lab imaging systems
Data from the FAME II trial has shown that patients with fractional flow reserve (FFR)-guided stenting plus the best available medical therapy had superior outcomes to those treated with medical therapy alone.
APPOSITION III is a prospective, single-arm, multicentre, post-market trial to assess the long term performance of the Stentys Self-Apposing stent in routine clinical practice in 1,000 STEMI patients.
CircuLite has announced that its Synergy Circulatory Support System will be included in four presentations at the 24th Annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium (Miami, USA, 22–26 October).
The four day training course offered hands-on training, analysis of cardiac CT data sets including post-percutaneous coronary intervention and surgery assessment skills.
Darby, who has 20 years of experience in the medical device industry, with most of those in the interventional cardiovascular space, was the business unit head of Bayer Interventional before joining Svelte.
The company specialises in the development of non-invasive technology for advanced haemodynamic monitoring, which can be used in the surgical, intensive care, emergency room and cardiology settings.
The company has been commercialising its CE-marked paclitaxel-coated drug-coated balloon for coronary (Primus) and peripheral (Legflow) applications since March 2012.
The results of the clinical trial with the Boston Scientific repositionable and retrievable device are expected to be used to support CE mark and other international regulatory approvals.
The study with the Revivent Myocardial Anchoring System for use in Less Invasive Ventricular Enhancement procedures commenced in Europe under the guidance of Louis Labrousse, Bordeaux-Pessac, France.
A scientific presentation and a live procedure of adjustment of the MiCardia device were presented by Martin Andreas and Markus Czesla to 200 cardiac surgeons and other professionals in Stuttgart, Germany.
The system gives healthcare professionals 24-hour access to digital inventory management, communities, eLearning, product information and technical support in order to reduce costs.
Results from the Argentine Registry of Acute Coronary Syndromes (SCAR) were presented by Ricardo Villarreal at the 38th Argentine Congress of Cardiology in Buenos Aires.
Under the agreement, Terumo will continue to incorporate BioMatrix in the production, marketing and selling of its drug"eluting stents in markets worldwide, outside of the United States.
The new study, published in the International Journal of Epidemiology, also found that sudden death was associated independently with poor educational attainment.
The study in Brisbane, Australia, is the first in which researchers examined the association between daily average temperature and "years of life lost" due to cardiovascular disease.
TRV027 has shown improvement in haemodynamics and has been well-tolerated in patients with stable advanced heart failure, supporting advancement into a phase 2b study in acute decompensated heart failure patients.
The draft quality standard on hypertension describes measurable markers of high-quality, cost-effective care to drive improvements in the effectiveness, safety and experience of care for people with hypertension.
The MediGuide Technology reduces the need for fluoroscopic images during cardiovascular procedures.
According to a study published in the European Journal of Heart Failure, severe aortic stenosis patients with low-mean gradient and reduced left ventricular ejection fraction (LVEF) have worse outcomes after transcatheter aortic valve implantation (TAVI) than patients with preserved LVEF and high-mean gradient.
Athanase Benetos, Nancy, France, and colleagues have found that higher blood pressure levels in very old patients do not predict mortality or major cardiovascular events but low pulse pressure amplification in this population does predict such events.
According to the company, the next-generation system will feature a new four electrode catheter that delivers radiofrequency energy simultaneously and is designed to significantly reduce ablation time during renal denervation procedures.
The announcement was made at a press conference in London by Boris Johnson, Mayor of London; Kevin Murphy, CEO of ExCeL London and Kim Fox, representing the ESC.
A team of researchers at the Erasmus University Medical Center, Rotterdam, The Netherlands, performed a cost-analysis of 84 propensity matched surgical and transcatheter aortic valve implantation patients. Ruben Osnabrugge, one of the authors of the study, writes for Cardiovascular News key conclusions of the analysis and compares it with the economic outcomes reported from the PARTNER trial.
New data presented at the PCR London Valves 2012 meeting has shown a 97% procedural success implantation with the CoreValve System (Medtronic) through a direct aortic approach.
The clinical trial is an open-label study that will randomise subjects 1:1 to renal denervation vs. no denervation with both groups receiving maximal tolerated doses of antihypertensive medications.
The CE-marked Carillon Mitral Contour System is designed to reduce functional mitral regurgitation while reducing mitral annulus dilatation upon device deployment.
ASSURE will evaluate the ability of RVX-208, Resverlogix's BET protein inhibitor, to regress atherosclerotic disease versus placebo using intravascular ultrasound (IVUS) technology in patients with coronary arterial disease.
The Emerge Catheter (Boston Scientific) is a next-generation pre-dilatation balloon catheter designed to treat challenging lesions in coronary arteries.
The new catheter to implant the Stentys Self-Apposing Stent adds a hydrophilic (slippery) coating and an ergonomic handle that considerably facilitates stent implantation in tortuous vessels.
According to Medtronic, the new valve incorporates technology that optimises fit, thereby promoting sealing between the prosthetic valve and the native valve.
Alain Cribier implanted the first transcatheter aortic valve in a patient at the University Hospital of Rouen, France, on 16 April 2002, he told Cardiovascular News how transcatheter aortic valve implantation (TAVI) began, how he dealt with criticism in the early days, complications with the procedure and the changes to the concept of the heart team.
A study published in the Journal of the American College of Cardiology, based on data from the Symplicity HTN-2 trial, suggests that renal denervation may be a cost-effective method of managing resistant hypertension and may result in a reduction in cardiovascular morbidity and mortality in the long term.
A large cohort study by Monique Anderson, Durham, USA, and colleagues have found that women experienced higher rates of all-cause in-hospital mortality and were more likely to experience a peri-procedural myocardial infarction, bleeding, and vascular complications than men undergoing percutaneous coronary intervention.
The international launch of Absorb, a first-of-its-kind device for the treatment of coronary arterial disease, is supported by five clinical studies in more than 20 countries around the world.
Affinity Fusion (Medtronic) is designed to serve as a patient's lungs by oxygenating and removing carbon dioxide from blood during various open-heart surgical procedures.
A study lead by researchers from the Mount Sinai Medical Center in Miami, USA, has found that patients with coronary valvular disease who were treated with a hybrid approach combining coronary artery stenting with minimally invasive valve surgery showed significantly better outcomes than patients who had standard sternotomy coronary bypass and valve surgery.
A study reported in JACC has shown successful implantation of the Tiara valve (Neovasc) in 81% of the test animals, with no obstruction of the left ventricular outflow tract, no pericardial effusion, no encroachment on the aortic valve, no transvalvular gradients and most importantly, no significant paravalvular leak.
CVInsight (Intelomed) displays vital information such as functional oxygen saturation of arterial haemoglobin (Sp02) and pulse rate of adult and paediatric patients.
A preclinical study and a first-in-man clinical study of the Primus Drug-Coated Balloon (Cardionovum) have shown "high procedural efficacy," and "no adverse clinical events observed out to six months."
BridgePoint Medical is the developer of an FDA cleared and CE marked catheter-based system designed to treat coronary chronic total occlusions.
The Perceval S valve (Sorin Group) is now approved for use in patients 65 years of age or older with aortic valve stenosis or steno-insufficiency.
The company plans to have a controlled launch with top heart hospitals in the United States, with full US commercial availability of the Impella CP expected by the fourth quarter of fiscal 2013.
Results of the CORE320 trial were presented during an ESC Hotline Session by João A C Lima, professor of Medicine and Radiology at the Johns Hopkins University School of Medicine.
Bernard Chevalier, interventional cardiologist, Cardiovascular Institute Paris-Sud, Jacques Cartier Hospital, Massy, France, talked to Cardiovascular News about his career, current research and innovations in interventional cardiology. He also spoke about a special interest in bioresorbable stent trials and his hobbies, including collecting fine wine.
The study conducted by scientists from the Johns Hopkins Children's Center, Baltimore, USA, and published online ahead of print in Circulation, can pave the way to new diagnostic tools and therapies for heart failure.
The VirtuoSaph Plus system provides an endoscopic approach to vessel harvesting, and is used for coronary artery and peripheral artery bypass graft procedures.
The Society for Cardiovascular Angiography and Interventions (SCAI) has launched the new SCAI Quality Improvement Toolkit (SCAI-QIT) Appropriate Use Criteria (AUC) and Guidelines app designed to provide information on coronary revascularisation appropriate use criteria.
The response includes information on reduction of risk of secondary cardiovascular events in patients with acute coronary syndrome.
Synergy is specifically targeted for treatment of ambulatory heart failure patients (INTERMACS 4,5,6 / NYHA IIIB and early IV) who remain symptomatic despite standard medical management.
The CORE320 study is a worldwide multicentre trial assessing the diagnostic performance of combined CTA and CTP as compared to traditional assessment of flow-limiting stenosis by means of SPECT-MPI and invasive coronary angiography.
The PROTECT study, which was presented at the ESC congress in Munich, failed to fulfil its hypothesis that the Endeavor zotarolimus-eluting stent would be superior to the Cypher sirolimus-eluting stent in terms of reducing the rate of definite or probable stent thrombosis.
The HERCULES trial found that patients with higher blood pressure levels at baseline had the most dramatic reduction in blood pressure following renal artery stenting with the RX Herculink Elite Stent (Abbott Vascular).
"We need to be very careful not to kill a promising technology with incorrect patient selection," Pierre-François Plouin, Paris, France, warned delegates at the EuroPCR Great Debate 2012 on renal denervation. In the debate, Plouin and four other experts in interventional cardiology, interventional radiology and nephrology agreed that identifying the right group of patients is key for the future of the technique.
The Elixir Desyne BD Novolimus Eluting Coronary Stent System was evaluated in the EXCELLA BD randomised clinical trial as compared to the control Endeavor Zotarolimus Eluting Coronary Stent System (Medtronic).
Results from the EnligHTN I trial showed that a reduction of 28 mmHg points systolic blood pressure seen after one month, remained stable at three months, using the EnligHTN renal denervation system.
If approved, the investigational device exemption (IDE) pivotal trial will study the C-Pulse System in patients suffering with moderate to severe heart failure.
More than 1,500 patients with severe aortic stenosis who are at high or extreme risk for aortic valve surgery have been included in the CoreValve US pivotal trial.
Cardiologists are restricting the use of transcatheter aortic valve implantation (TAVI) to very old or very sick patients at high surgical risk, according to results of the Transcatheter Valve Treatment (TCVT) Sentinel Pilot registry presented at the ESC Congress.
First results from the German Aortic Valve Registry (GARY) show that participating centres using transcatheter aortic valve implantation (TAVI) are adhering to current guidelines for patient selection.
The device's regulatory and reimbursement approvals earlier this year were supported by the results of RESOLUTE Japan, a single-arm study that enrolled 100 patients at 14 Japanese centres.
Results from the DeFACTO study assessing the performance of non-invasive fractional flow reserve (FFRCT) technology were presented at ESC and published simultaneously in JAMA
Following an investigation of the ambulance records in the area close to the epicentre of the earthquake investigators found that the occurrence of cardiovascular conditions increased sharply after the earthquake.
Instances of hospital re-admission leading to urgent revascularisation were reduced by 86% for patients in the FFR-guided percutaneous coronary intervention arm of the trial.
Ticagrelor received a class I recommendation from the ESC in the revised Guidelines on the Management of Acute Myocardial Infarction in Patients Presenting with STEMI.
Largest prospective randomised drug-eluting stent trial ever presented, ESC late-breaker on the PROTECT study reveals low rates of stent thrombosis through three years of follow-up.
A novel non-invasive device which separates healthy and damaged heart muscle and restores ventricle function improves three-year outcomes in patients with ischaemic heart failure.
An eighteen-month update from the SYMPLICITY HTN-2 randomised clinical trial was presented at ESC Congress 2012 on 26 August 2012 in Munich, Germany.
Philips becomes the exclusive distributor of Corindus' CorPath 200 System, the world's first robotic-assisted system for the minimally invasive treatment of obstructed coronary arteries.
According to Toshiba, its new 16-detector row CT system provides faster imaging reconstruction and helps lower radiation dose for safer patient exams.
The appointment of Noël Coopmans followed the introduction of Blue Medical's new drug-eluting technologies in March of this year and the development of additional coronary and peripheral applications.
The European Society of Cardiology board has approved a change in the name of the European Association of Echocardiography (EAE) to European Association of Cardiovascular Imaging (EACVI). Luigi Badano, EAE president, spoke to Cardiovascular News about this change.
Stephen Westaby and David Taggart, Department of Cardiothoracic Surgery, John Radcliffe Hospital, Oxford, UK, published an editorial in Heart criticising the fact that UK's National Health Service does not fund circulatory support equipment to treat cardiogenic shock in the majority of cardiac tertiary centres.
The 90-patient, placebo-controlled, randomised double-blinded phase II study is evaluating the safety and efficacy for JVS-100 in patients with late stage heart failure.
The approval includes expanded CE mark indications to treat patients with complex coronary arterial disease and a minimum duration of three months of dual anti-platelet therapy.
The multicentre randomised trial of MGuard (InspireMD) demonstrated a positive outcome in treating patients suffering acute myocardial infarction when compared to commercially-approved bare metal or drug-eluting stents.
Venture is a deflectable-tip catheter used to provide guidewire directional control in challenging coronary and peripheral interventional procedures.
FMwand's precise control of thermal energy minimises damage to tissue surrounding the incision, preserving healthier, viable tissue.
The manufacturer, Mylan Pharmaceuticals, gained FDA approval for 15mg, 30mg and 45mg pioglitazone tablets.
Researchers from the Peninsula College of Medicine and Dentistry (PCMD), University of Exeter, and University of Cambridge have for the first time established a link between high levels of urinary Bisphenol-A (BPA) and severe coronary artery stenosis.
Retia's cardiovascular monitor is a less-invasive system to monitor cardiac output and other key measures of the cardiovascular system accurately when patients go into shock.
The Jarvik 2000 is used to provide long-term, permanent support to end-stage congestive heart failure patients who are not candidates for heart transplant.
"A strategy incorporating measurable anatomical and procedural variables together with post-implantation corrective measures can reduce the incidence of paravalvular aortic regurgitation after transfemoral TAVI," says Mohamed Abdel-Wahab, consultant cardiologist, Heart Center, Segeberger Kliniken, Bad Segeberg, Germany.
On 15 August, the UK's National Institute for Health and Clinical Excellence (NICE) opened a consultation on its draft medical technology guidance on the use of WatchBP Home A (Microlife), a device that measures blood pressure and detects atrial fibrillation.
Three-month results from the SOUND-ITV study have shown that the catheter-based ultrasound system from Sound Interventions significantly lower blood pressure in patients whose blood pressure was unable to be controlled by conventional pharmaceutical therapy.
A study published in the current issue of the New England Journal of Medicine (NEJM) has shown that survival using the Excor Pediatric Ventricular Assist Device (VAD) (Berlin Heart) is significantly greater than the standard-of-care (extracorporeal membrane oxygenation, or ECMO) as a bridging therapy for children in need of a heart transplant.
The new Prolimus Biograde DES technology promotes early re-endothelialisation and rapid healing.
The purpose of the post approval study for the Excor paediatric VAD (Berlin Heart) is to evaluate whether safety and outcomes of the device in the commercial setting are comparable to the safety and outcomes shown in the IDE study.
The case is part of the ARTDIVA (Arterial remodeling transient dismantling vascular angioplasty) clinical trial.
Binita Shah, New York University School of Medicine, New York, USA, explains the results of a study that demonstrated that even in the hands of experienced operators, radiation exposure is higher when using transradial approach compared with transfemoral approach.
Robert M A van der Boon, Erasmus Medical Center, Rotterdam, The Netherlands, writes that the possibility that conduction abnormalities may resolve early or late after TAVI puts the clinical implications of permanent pacemaker implantation after TAVI in a different perspective.
Corindus Vascular Robotics has received FDA 510(k) clearance for its CorPath 200 System to be used in percutaneous coronary intervention (PCI) procedures. Results of the PRECISE trial showed that PCI with the system was successfully completed without having to convert to manual percutaneous coronary intervention in 98.8% of patients and without device-related complications.
The trial, called SURTAVI (Medtronic CoreValve surgical replacement and transcatheter aortic valve implantation) trial, will evaluate the potential for the minimally-invasive CoreValve system to be considered for less-sick patients who typically are treated with open-heart surgical aortic valve replacement today.
Glider (TriReme) is now approved for the treatment of lesions in the coronary vasculature in balloon diameters from 1.5 – 3.5mm and in lengths from 4mm to 20mm.
Rcadia Medical Imaging announced results of an initial study intended to demonstrate the feasibility of a proprietary technique that uses coronary CT angiography studies (cCTA) as a non-invasive alternative to assess the haemodynamic significance of coronary lesions.
Janssen R&D has announced it is withdrawing the supplemental new drug application (sNDA) to the FDA for the use of rivaroxaban (Xarelto, Bayer/Janssen) to reduce the risk of stent thrombosis in patients with acute coronary syndrome (ACS).
The treatments are part of the company's ORBIT II trial, which is now over 70% enrolled.
The TRANSFORM trial is the first US clinical trial of a rapid deployment system for surgical aortic valve replacement procedures.
On 2 July, CVRx launched a European post-market hypertension registry to track performance outcomes and physician experience using the company's Barostim neo device for hypertension. The registry is expected to enrol up to 500 patients.
The Somatom Definition Edge CT system offers high image quality, dose reduction and dual energy in clinical settings, including the emergency department.
The CE mark includes a transaortic indication for delivery of the valve through mini-sternotomy or mini-thoracotomy, after which the valve is then implanted through a small incision in the aorta.
John Ormiston, Auckland, New Zealand, has investigated important innovations in interventional cardiology such as bioresorbable stents, TAVI and renal denervation. He spoke to Cardiovascular News on these technologies.
RE-ALIGN is designed to evaluate dabigatran etexilate (Pradaxa, Boehringer Ingelheim) as an alternative to warfarin for use in patients with mechanical heart valves requiring anticoagulation therapy.
The cost-effectiveness of ticagrelor (AstraZeneca) was driven by the mortality benefit seen in the pivotal PLATO trial, which showed ticagrelor was cost-effective across all major patient subgroups.
The Transcatheter Mitral Valve Implantation (TMVI) procedure was performed on 12 June 2012, at The Heart Centre, Rigshospitalet University Hospital, Copenhagen, Denmark.
The randomised study will enrol 150 heart attack patients who will be treated with either the Stentys stent or Medtronic's Resolute drug-eluting stent.
The FDA Circulatory System Devices Panel voted unanimously, with one abstention, that TAVI with the Edwards Sapien device should be approved for use in patient considered high-risk for surgery.
The first-in-man experience with the Claret Pro cerebral protection system showed that more than half of the patients had debris captured during transcatheter valve implantation.
Results of PRECISE, a multicentre, pivotal investigation with the CorPath 200 robotic system, were presented at a late-breaking clinical trial at the SCAI annual meeting in Las Vegas.
Edwards Lifesciences announced that it has received conditional Investigational Device Exemption (IDE) approval from the FDA to initiate a clinical trial to study its GLX next-generation tissue treatment platform applied to a surgical bovine pericardial heart valve.
The ESC has announced an update to their guidelines for the management of chronic heart failure highlighting the crucial role of mineralocorticoid receptor antagonists (MRA).
Across four different dosing regimens tested, patients receiving SAR236553/REGN727 for 12 weeks achieved a mean LDL-cholesterol reduction from baseline of 28.9% to 67.9%, compared to 10.7% in patients receiving placebo.
At two years of follow-up in the RESOLUTE clinical study, rates of target lesion failure are 7.1% for patients without diabetes and 9.6% for patients with diabetes.
Results of ADVANCE a US clinical trial evaluating the use of the HeartWare Ventricular Assist System (HeartWare International) as a bridge to heart transplantation in patients with advanced heart failure were published on 22 May ahead of print in Circulation.
The FDA's Cardiovascular and Renal Drugs advisory committee voted six to four against rivaroxaban's new indication based on missing data on early patient withdrawals from the phase 3 ATLAS ACS 2 TIMI 51 study.
Two new denervation systems were launched at EuroPCR (Paris, 15–18 May 2012). Covidien unveiled its OneShot system and St Jude Medical announced the launch of the EnligHTN system.
Six-month results from the DELUX registry demonstrated positive safety and efficacy outcomes for the Pantera Lux Paclitaxel Releasing Balloon (Biotronik) in more than 1000 patients.
Results from 31 treated US and European patients demonstrate a near-full New York Heart Association (NYHA) class improvement at two years compared to baseline, also a reduction in left ventricular volume was maintained.
Two-year clinical data shows low adverse event rates, including no myocardial infarction or stent thrombosis.
The Stentys AC thrombectomy device is designed to safely recover debris and can be used in concert with its Self-Apposing devices, Stentys BMS and Stentys DES.
Lead investigator, Dariusz Dudek, Jagiellonian University, Krakow, Poland, presented positive three-year results from the extended follow-up MAGICAL trial at a InspireMD-sponsored symposium at EuroPCR.
Data from the CoreValve ADVANCE study found that survival rates were nearly identical between genders, with no statistical differences in 30-day and 6-month all-cause mortality, cardiovascular mortality or the 30-day MACCE endpoint.
Thirty-day outcomes for patients enrolled in the SOURCE XT Registry demonstrated low all-cause mortality of 6.3% and low rates of procedural complications.
Data shows statistically significant advantages of using an IN.PACT drug-eluting balloon (Medtronic) over a corresponding conventional treatment for coronary and peripheral artery disease, respectively.
The Synergy stent (Boston Scientific) shows low rates of revascularisation and no cardiac-related deaths or stent thrombosis at one year.
ENERGY is a registry in a broad patient population investigating the safety and clinical performance of the PRO-Kinetic Energy bare metal stent system (Boston Scientific) in 1,016 patients.
The Lotus Aortic Valve System (Boston Scientific) showed successful deployment in all patients (11) with virtually no paravalvular regurgitation after valve placement or at discharge.
The Resolute Integrity DES (Medtronic) is now indicated in countries approving the CE mark for the following patient and lesiontypes: acute coronary syndromes, acute myocardial infarction, unstable angina; diabetes mellitus; bifurcations; multi-vessel disease; total occlusions and chronic total occlusions; and in-stent restenosis. All but the indications for diabetes and multi-vessel disease are new.
Michael Haude told EuroPCR delegates that in-stent and in-segment late loss and binary restenosis rates were low for Combo and comparable to those of Taxus; Combo also showed a uniform, homogeneous neointimal response.
Clinical data from the EnligHTN I trial about the safety and efficacy of the company's renal denervation technology will be presented on Wednesday 16 May.
Three-month dual anti-platelet therapy is the shortest duration for any major drug-eluting stent in Europe.
Neovasc has announced that its Tiara technology under development for the transcatheter treatment of mitral valve disease will be featured in three scientific sessions at EuroPCR 2012 (Paris, France, 15–18 May).
The FDA has cleared the Mega 7.5Fr 30cc and 40cc intra-aortic balloon catheters. These larger volumes provide patients with greater haemodynamic support, regardless of their height.
Boston Scientific and Philips Healthcare have announced a collaboration enabling Philips to sell Boston Scientific Imaging equipment in connection with its Allura Xper catheterisation (cath) lab systems throughout the world.
Cardionovum has announced that it will launch two drug-eluting balloon (DEB) dilatation catheter product families at EuroPCR 2012 congress: Paclitaxel-coated drug-eluting balloons for coronary (Primus) and peripheral (Legflow) applications.
The True Dilatation Balloon Valvuloplasty Catheter (Loma Vista) is specifically designed to prevent balloon ruptures in balloon aortic valvuloplasty and transcatheter aortic valve implantation procedures.
Results of the multicentre PRECISE study show effectiveness of a robotic-assisted system for patients and enhanced safety for interventional cardiologists performing percutaneous coronary interventions.
The new drug application seeks approval to reduce the risk of stent thrombosis in patients with acute coronary syndrome.
The trial will evaluate Promus Element platinum-chromium everolimus-eluting stents versus Omega bare-metal stents in patients with heart attack undergoing primary angioplasty with bivalirudin anticoagulation.
Hitachi Aloka Medical announced its collaboration with Alan Fraser of the Cardiff University School of Medicine to establish industry standards for the development and evaluation of diagnostic tools that examine how the heart interacts with larger arteries.
Cardio3 BioSciences has announced that it has received CE mark for its intra-myocardial C-Cath Injection Catheter.
The results of the study, published in the April 2012 edition of Eurointervention, also showed that patients using atorvastatin had a 23% lower mortality than those using simvastatin.
The new European Society of Cardiology (ESC) guidelines, launched at EuroPrevent 2012, stress that cardiovascular disease prevention should be a life-long effort that starts in the womb and lasts to the end of life.
Patients enrolled in the SOUND-ITV study will be followed for 12 months to evaluate the procedure's effectiveness in lowering blood pressure.
New data from the TRITON trial show that the Edwards Intuity valve system facilitates small-incision surgery for aortic valve replacement, with a high procedural success rate, and consistent and sustained haemodynamic valve performance at one year.
According to GE Healhcare, the system provides innovative tools designed to help improve workflow efficiency through simplified image acquisition, intuitive navigation and advanced yet easy to use quantification.
Producing high-quality clinical images and reducing radiation exposure with Adaptive Iterative Dose Reduction 3D (AIDR 3D), the system can generate 80 unique slices per rotation.
Edwards Lifesciences announced that, effective immediately, the CMS will reimburse TAVI procedures for US patients with symptomatic aortic stenosis under specified criteria.
Recent data from commercial use of the device in 41 patients at St Marien Hospital, Siegen, Germany, showed 100% success rate and no complications.
Post-PASSION trial analysis published in the Catheterization and Cardiovascular Interventions journal confirms no difference in terms of major adverse cardiac events.
ThruPort IntraClude is indicated for use in patients undergoing cardiopulmonary bypass, including during mitral heart valve repair or replacement surgery using a minimally invasive approach.
The V2 System is an over-the-wire balloon catheter with an array of electrodes mounted in a precise pattern designed to deliver a modest dose of radiofrequency energy to disrupt the renal nerves.
DNA from the heart's own cells plays a role in heart failure by mistakenly activating the body's immune system, research co-funded by the British Heart Foundation has found.
Data presented on 27 April at the 22nd annual scientific meeting of the European Society of Hypertension showed patients (n=84) who received renal denervation treatment with Symplicity experienced a mean blood pressure reduction of -28/-10 mm Hg (p<0.001) at six months following treatment compared with baseline.
The Promus Element Plus Everolimus-Eluting Platinum Chromium Coronary Stent System (Boston Scientific) has received Health Canada approval for the treatment of patients with coronary artery disease.
The Emerge catheter (Boston Scientific) includes a 1.2mm diameter balloon option that features an ultra-low 0.017" tip profile and low crossing profiles designed to cross tight coronary lesions.
In the study, researchers followed 50 Japanese patients who received 84 Igaki-Tamai stents to treat 63 lesions between September 1998 and April 2000.
The results of the prospective, single-arm feasibility REPRISE I trial, which enrolled 11 patients at three sites in Australia, will be presented at EuroPCR in Paris.
At three months, patients who received cell therapy experienced an 11 point improvement in the ejection fraction in comparison with the control group.
The valve is constructed using a polyester and tissue-covered, fatigue resistant, high strength titanium stent to ensure structural integrity of the valve.
The Impella cVAD1 is designed to provide temporary circulatory support via a minimally invasive, catheter-based pump that is inserted percutaneously, without the need for surgical intervention.
SynCardia Systems announced that the FDA has approved a humanitarian use device designation for its temporary Total Artificial Heart to be used for destination therapy in addition to its current premarket approval as a bridge to transplant.
Approval of Xience Prime in Japan was supported by results from the SPIRIT PRIME clinical trial, a prospective, open-label trial that evaluated Xience Prime in approximately 500 patients with coronary artery disease.
The main updates are recommendations on the use of transcatheter aortic valve implantation (TAVI) for people who would be considered unsuitable for open heart surgery.
Data from REMEDEE demonstrate the safety and feasibility of the Genous stent (OrbusNeich) for patients with ST-segment elevation myocardial infarction (STEMI) and showed no incidence of very late stent thrombosis.
A meta-analysis published online in The Lancet and presented at the American College of Cardiology's (ACC) 61st Annual Scientific Session in Chicago, USA, shows that cobalt-chromium (Co-Cr) everolimus-eluting stents (Xience, Abbott Vascular) have much lower short-term and long-term stent thrombosis rates than both bare metal stents and all other drug-eluting stents.
Two-year follow-up of patients in the PARTNER trial supports the use of transcatheter aortic valve implantation (TAVI) as an alternative to surgery in selected high-risk patients with aortic stenosis.
James White, London, Canada, writes about the first evaluation of diagnostic utility for cardiac imaging in patients presenting with ventricular arrhythmias.
A statistically significant reduction in recurrent ischaemia requiring repeat revascularisation was present between the first and second year of follow-up with the platinum chromium stent.
Results from the Balloon Pump-Assisted Coronary Intervention Study (BCIS-1) were presented at the American College of Cardiology's 61st Annual Scientific Session.
Results of the ADVANCE study showed that survival rates were high at both 30 days (95.5%) and six months (87.2%) in patients with severe aortic stenosis who are at high-risk for surgery.
Data from two clinical trials presented at the 61st Annual Scientific Session of the American College of Cardiology show consistent effectiveness and safety up to three years.
Recently FDA-approved, device shows consistently low event rates across broad spectrum of coronary artery disease patients, including those with diabetes.
The PressureWire with Agile Tip was designed to replicate the performance of standard percutaneous coronary intervention guidewires and deliver exceptional handling even in the most challenging anatomies.
The VectraplexECG System uses only five electrodes to detect ECG changes suggestive of acute myorcardial infarction and derive a 15-lead ECG.
After seven years of follow-up, depressed patients were 1.5 times more likely to have died than non-depressed patients, research presented at 12th Annual Spring Meeting on Cardiovascular Nursing has shown.
The randomised trial has concluded that a catheter procedure to close a patent foramen ovale is no more effective than medical therapy in preventing recurrent strokes.
German sub-study from the PARACHUTE Cohort B trial continued to show positive results after heart failure patients were treated with the Parachute device.
The phase II trial of JVS-100 will evaluate the safety and efficacy of Stromal-cell Derived Factor 1 (SDF-1)─a strong chemo-attractant of stem cells and progenitor cells that promote tissue preservation and increased and sustained healing of wounded tissue in heart failure patients.
"RenalGuard (PLC) offers us a potential method to reduce the incidence of contrast-induced nephropathy, a major issue when performing interventional procedures requiring contrast agents with high-risk patients," said Eugenia Nikolsky, Rambam Medical Center, Haifa, Israel.
Catheter-based mitral valve repair with MitraClip (Abbott Vascular) is the best-studied type of percutaneous mitral valve therapy, said Ted Feldman, Evanston, USA, at the International Symposium on Endovascular Therapy (ISET) in Miami.
A retrospective analysis conducted in Germany shows that fluoroscopy times and radiation exposure in patients undergoing renal denervation are similar to those in patients undergoing routine diagnostic catheterisation with elective percutaneous coronary intervention but lower than in patients undergoing emergency intervention.
The addition of computed tomography (CT) perfusion to CT angiography improves diagnostic accuracy of CT angiography in acute chest pain patients, a study presented at the European Congress of Radiology 2012 in Vienna, Austria, shows.
Marie-Claude Morice, Massy, France, analyses the results of a single-centre study comparing the outcomes of transcatheter aortic valve implantation in men and women.
The Nuloy coronary stent (ICON Interventional Systems) has extremely thin struts, superior flexibility, improved deliverability and lower profile.
Data from the recently published GENIA study show that the Genous stent (OrbusNeich) is associated with low rates of stent thrombosis and target lesion revascularisation.
The RAMSES trial is designed to understand how Picso can help acute coronary syndrome patients with large anterior infarcts having ECG changes with poor microcirculation after revascularisation.
The Promus Element stent system incorporates a platinum chromium (PtCr) alloy with an innovative stent design and an advanced catheter delivery system designed to provide physicians improved drug-eluting stent performance in treating patients with coronary artery disease.
The trial, sponsored by Baxter, will enrol approximately 450 patients across 50 clinical sites in the United States, who will be randomised to one of three arms: treatment with their own autologous CD34+ stem cells, treatment with placebo (control), or unblinded standard of care.
This decision was based upon data from the global Phase III ATLAS ACS 2-TIMI 51 study, which showed that rivaroxaban significantly reduced the composite of cardiovascular death, myocardial infarction and stroke, versus placebo.
The treatment with the Dior device resulted in low major adverse cardiac events, target lesion revascularisation and target vessel revascularisation rates according to Antonio Serra, Barcelona, Spain.
The US Food and Drug Administration has announced safety changes to the labeling for some widely used statins.
Sachin Goel and Samir Kapadia, Cleveland Clinic, USA, say that PCI can be performed in severe aortic stenosis patients and coronary artery disease without an increase in short-term mortality or procedural complications.
REDUCE-HTN is a non-randomised, prospective, 64-patient clinical study designed to validate the clinical performance of the Vessix system for medication-resistant hypertensive patients.
Ion and Taxus Liberte paclitaxel-eluting stent systems are now approved for treating patients with acute myocardial infarction in the USA.
The hydrogel is made from cardiac tissue that is stripped of heart muscle cells through, freeze-dried and milled into powder form, and then liquefied into a fluid that can be injected into the heart.
The first patient was enrolled in the Freedom Solo IDE study at St Vincent's Heart Center of Indiana in Indianapolis by David Heimansohn, the study's principal investigator.
DSM will contribute its technology to enable UMC Utrecht to develop and evaluate a prototype of a non-biological supportive scaffold for the minimally invasive treatment of valvular and vascular diseases.
The new oral antiplatelet drug is now included in seven sets of cardiovascular treatment guidelines across the world, two sets in the EU, four in the USA and one in Canada.
Paradise (ReCor Medical) denervates all the way around the arterial wall while simultaneously cooling the endothelium, to help enable a safe, consistent, and fast renal denervation procedure.
The approval stems from the results of a series of studies involving the Medtronic device, which showed powerful results across a broad spectrum of patients, including those with diabetes.
The system, which consists of a bovine pericardial heart valve and novel delivery system, facilitates small incision surgery and rapid valve deployment during aortic valve replacement procedures.
Country-specific analysis will evaluate the cost effectiveness of an FFR-guided intervention strategy for patients with multivessel coronary artery disease in Japan, China, India, Korea, and Australia.
The American Heart Association has published a statement, online in Circulation: Journal of the American Heart Association, containing recommendations on sexual activity for patients with stable cardiovascular disease.
Experience and improved patient selection have improved outcomes with transapical aortic valve implantation in the PARTNER trial Non-Randomized Continued Access (NRCA) programme, new results have shown.
Jeffrey Moses, New York, USA, notes that percutaneous aortic valve implantation is probably the most transformative technology in the last 20 years and describes his father's experience of undergoing a TAVI procedure, in this interview with Cardiovascular News. He also spoke about his career, influences, areas of research and his love for skiing and playing tennis.
Christian J Terkelsen, Department of Cardiology, Aarhus University Hospital, Skejby, Denmark, writes about the country's reperfusion strategy and the importance of prehospital diagnosis and field-triage.
Follow-up trial to landmark FAME trial examines benefits of using FFR-guided assessment for stent procedures with optimal medical treatment compared to optimal medical treatment alone.
Manuel Castella, Barcelona, Spain, was a principal investigator in the FAST trial, he gives his insights into the design and outcomes of this trial that set out to compare endocardial catheter ablation isolation of the pulmonary veins vs. minimally invasive thoracoscopic surgical epicardial isolation for atrial fibrillation treatment.
The IMPROVE HF bridge study will examine specific gaps in implementation of guideline-recommended treatment in heart failure patients post-hospital discharge.
The Promus Element Plus stent system employs an advanced low-profile delivery system featuring a dual-layer balloon designed to enable precise stent delivery across challenging lesions and reduce balloon growth during inflation to facilitate high-pressure stent deployment in treating patients with coronary artery disease.
APPOSITION III is a multicentre, prospective, observational registry that seeks to evaluate the long term safety and performance of the Stentys Self-Apposing coronary stent to treat acute myocardial infarction.
The Parachute is a catheter based partitioning device deployed within the left ventricle for patients who have developed ischaemic heart failure following a heart attack.
The UK's National Institute for Health and Clinical Excellence (NICE) has recommended the Discovery CT750 HD scanner as an option for first line cardiac imaging of the coronary arteries in people with suspected coronary artery disease and for 'difficult to image' patients.
Data presented at the Society of Thoracic Surgeons (STS) 48th Annual Meeting (Fort Lauderdale, USA, 28 January–1 February) demonstrate positive outcomes when the CoreValve system (Medtronic) is implanted using the direct aortic approach.
Medtronic will initially distribute Miami Instruments' Joseph Lamelas knot pusher and the Joseph Lamelas atrial lift system used during minimally invasive cardiac surgery procedures.
The Freedom portable driver (Syncardia Systems) allows stable Syncardia's Total Artificial Heart patients to be discharged from the hospital while they wait for a matching donor heart.
The study is a multicentre, randomised, double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of infarct-related artery infusion of AMR-001, an autologous bone marrow derived cell therapy enriched for CD34+ cells.
A study published in the Journal of the American Medical Association (JAMA) shows that the novel drug cangrelor (The Medicines Company) is associated with a higher rate of platelet inhibition, compared with placebo, in patients who have discontinued thienopyridine therapy prior to cardiac surgery.
For the first time, the St George's Valve Technology Symposium now in its seventh year will be an integral part of the Charing Cross International Symposium. Now called the CX St George's Valve Technology Symposium, the event will take place during the first two days of the 34th CX Symposium (14–17 April 2012, Imperial College London, UK).
Percutaneous coronary intervention with a drug-eluting balloon for in-stent restenosis is a promising effective therapeutic option with an unexpected low repeat restenosis and target lesion revascularisation rate, according to research presented at the American Heart Association's Scientific Sessions 2011.
Olaf Wendler, London, UK, summarises and comments on one-year outcomes from the SOURCE registry. SOURCE provided procedural results with the commercialised Edwards Sapien valve.
Stem cells from the placenta can travel to injured tissues in a mother's heart during pregnancy and create new cardiac cells, according to a late-breaking basic science abstract presented at the American Heart Association's Scientific Sessions 2011.
Results from the BRIDGE trial showed 99% of cangrelor-treated patients maintained target levels of platelet inhibition for all time points measured over the bridging period to cardiac surgery compared to 19% of placebo patients with no significant excess in surgical bleeding complications.
The study will further evaluate the safety and effectiveness of the InnerCool RTx endovascular cooling system (Phillips) to reduce reperfusion injury in acute ST-elevation myocardial infarction (STEMI) patients.
Techulon's polymers were used as delivery agents for an NF-κB oligodeoxynucleotide (ODN) decoy used to limit myocardial infarction during an ischaemic-repurfusion event simulating a heart attack.
Results were taken from a 12-month follow-up post-hoc analysis of e-HEALING, a multicentre, worldwide prospective clinical registry conducted in 31 countries outside of the United States that included 4,939 patients.
The Cardiovascular Medical Research and Education Fund (CMREF) has awarded CircuLite approximately US$950,000 to support ongoing work to modify the design of its existing Synergy system so that it can be used to provide circulatory support for a failing right heart.
The Bioabsorbable Cardiac Matrix (Ikaria) an aqueous mixture of sodium alginate and calcium gluconate is being investigated to prevent ventricular remodeling and subsequent congestive heart failure following acute myocardial infarction.
The filing is supported by data from the pivotal phase III ATLAS ACS 2 TIMI 51trial presented in November at the American Heart Association Scientific Sessions.
The system is intended to bridge paediatric patients from newborns to teenagers awaiting heart transplantation from days up to several months, until a donor heart becomes available.
This first implant is part of St Jude's European clinical trial to assess the safety and effectiveness of the Portico heart valve for patients with severe symptomatic aortic stenosis, who are at high risk for conventional open-heart valve replacement therapy.
This is the case study of a patient with non-ST elevation myocardial infarction (NSTEMI), which shows successful clinical outcomes at 12-month follow-up post Genous stent implantation after only one month of dual antiplatelet therapy (DAPT).
Sven Plein, Leeds, UK spoke to Cardiovascular News about the current trends in cardiovascular magnetic resonance (CMR), the benefits of this modality and the recent use of CMR as an endpoint in acute myocardial infarction trials.
Amar Krishnaswamy and Murat Tuzcu, Cleveland Clinic, USA, speak on the resurgence of balloon aortic valvuloplasty as a treatment to bridge severely symptomatic patients to transcatheter or surgical aortic valve replacement, or to better understand the contribution of aortic stenosis to functional limitation in patients with multiple comorbidities.
Approximately 500 patients with coronary artery disease will be enrolled at about 40 investigational sites in Europe and New Zealand to evaluate the Absorb BSV compared to a metallic drug-eluting stent.
The GENE-MATRIX study will determine optimal antiplatelet therapy with the Spartan RX CYP2C19 point-of-care DNA testing system which will be used to test heart attack patients receiving cardiac stents.
Data from a multicentre trial show that the Genous stent (OrbusNeich) is safe when accompanied by only 10 days of dual antiplatelet therapy.
Four-year results from the LEADERS trial show that BioMatrix Flex, Biosensors' Biolimus A9-eluting stent system with a biodegradable polymer coating, significantly reduces the risk of very late stent thrombosis compared to that of a drug-eluting stent system with a durable polymer coating.
The eSVS Mesh (Kips Bay Medical) is a nitinol mesh sleeve that, when placed over a saphenous vein graft during CABG surgery, is designed to improve the structural characteristics and long-term performance of the vein graft.
The Promus Element stent (Boston Scientific) uses a proprietary PtCr alloy designed specifically for coronary stenting, which enables thinner struts and enhanced visibility.
Ulrich Sigwart, Switzerland, is one of the pioneers in coronary angioplasty, having performed his first case in 1978. He told Cardiovascular News about this first experience, his most memorable cases, and why he considers himself a "nomad".
The PARTNER Trial Cohort B results demonstrate widening survival benefit for transcatheter aortic valve implantation (TAVI) patients at two years, with mortality for TAVI at 43.4% compared to 67.6% for medical therapy. These new results were presented by Raj Makkar, USA, at the 23rd annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium, San Francisco, USA.
A novel technology to measure stenosis severity yields similar results to traditional fractional flow reserve (FFR) without the use of adenosine to induce maximum hyperemia.
At five years, there were no reports of cardiac deaths, blood clots or ischaemia-driven target lesion revascularisation in the first stage of the ABSORB trial.
At 30 days, study results from the Portico transcatheter aortic heart valve (St Jude Medical) showed no device or procedure-related adverse events or death.
The Combo stent, the worlds' first dual therapy stent (DTS), combines an abluminal biodegradable sirolimus elution for the regulation of hyperplasia with Genous, OrbusNeich's proven endothelial progenitor cell (EPC) capture technology, to accelerate re-endothelialisation.
"The Synergy stent (Boston Scientific) met the primary non-inferiority endpoint of six-month late loss compared to the Promus Element stent (Boston Scientific), demonstrating effectiveness of drug elution from this abluminal bioabsorbable polymer," said Ian T Meredith, principal investigator of the trial.
OrbusNeich announced that three-year clinical outcomes data from the use of the Genous stent for percutaneous coronary interventions in a large, real-world patient population demonstrate the device's long-term safety and efficacy.
Six-month data from the DELUX registry and 12-month outcomes from the PEPPER study, both demonstrated favourable safety and efficacy results for the Pantera Lux paclitaxel releasing balloon (Biotronik).
John Ormiston, Auckland, New Zealand, presented latest data from the first-in-human DESSOLVE I trial of the MiStent Sirolimus drug-eluting coronary stent system (Micell Technologies) at the Transcatheter Cardiovascular Therapeutics Conference (TCT) 2011, in San Francisco, USA.
The PLATINUM Long Lesion study met its primary endpoint of target lesion failure at 12 months with a 3.2% rate for the Promus Element (Boston Scientific) stent in the per protocol population compared to a pre-specified performance goal of 19.4%.
The Genous stent (OrbusNeich) had 69.7% healed and 5.3% malapposed struts compared to 56.7% healed and 19.7% malapposed struts for the Blazer stent (OrbusNeich), 55.2% healed and 13.9% malapposed struts for the SolarFlex stent (Disa Vascular) and 34% healed and 45.7% malapposed struts for the Nobori stent (Terumo).
"The excellent clinical results at two years confirm the ION (Boston Scientific) stent's safety and efficacy in workhorse lesions and suggest superior performance relative to bare-metal stents in small vessels," said Louis Cannon, USA, principal investigator of the trial.
According to Medtronic, early evaluations have shown the direct aortic approach demonstrates high procedural success rates and high overall survival rates that are similar to those from transfemoral and subclavian approaches.
Latest data on the MitraClip system for mitral valve repair, the Absorb bioresorbable vascular scaffold for the treatment of coronay artery disease, Xience Prime everolimus eluting coronary stent system and Xience V will be presented at the 23rd annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium (7 – 11 November, San Francisco, USA).
Edwards Lifesciences has received approval for the transfemoral delivery of the Sapien device for the treatment of inoperable patients with severe symptomatic aortic stenosis.
The trial will evaluate the safety and effectiveness of Medtronic's CoreValve system in patients with symptomatic, severe aortic stenosis who are at high risk, or are ineligible, for open-heart surgery.
The Gore REDUCE clinical study is designed to demonstrate the safety and effectiveness of Gore Helex Septal Occluder for patent foramen ovale closure in patients with a history of cryptogenic stroke.
FDA approval of Xience Prime was supported by results from the SPIRIT PRIME clinical trial, a prospective, open-label study that met its primary endpoint with low rates of target lesion failure and stent thrombosis at one year.
Twelve-month follow-up data from the e-HEALING (Healthy endothelial accelerated lining inhibits neointimal growth) registry shows that the Genous stent (OrbusNeich) is safe and effective in elderly patients. The study was published in Circulation.
The combined fractional flow reserve (FFR) and optical coherence tomography (OCT) system from St Jude Medical offers physicians advanced physiological and anatomical insight to improve the diagnosis and treatment of coronary artery disease.
NeoStem, intends to initiate, no later than the first quarter of 2012, a phase II clinical trial for Amorcyte's lead product candidate, AMR-001, for the treatment of acute myocardial infarction.
Transcatheter valve implantation (TAVI) penetration rates are under 20% in Europe, according to data presented on 17 October at the PCR London Valves congress in London, UK.
Giuseppe De Luca, Novara, Italy, comments on the results of the DESSERT (Drug-eluting stents in primary angioplasty) cooperation, a meta-analysis of randomised trials which aimed to evaluate the risks and benefits of drug-eluting stents in comparison to bare metal stents in patients undergoing primary percutaneous coronary intervention for STEMI.
Intellectual medical property from NMT includes the data related to CLOSURE I, a trial that may create better clinical understanding of patent foramen ovale in stroke patients.
The Promus Element represents Boston Scientific's third-generation drug-eluting stent (DES) technology, which incorporates a novel platinum chromium (PtCr) alloy, innovative stent design and advanced catheter delivery system to treat coronary artery disease.
"During the six-month follow-up period of the APPOSITION I study, there was no death, no stent thrombosis or reoccurrence of myocardial infarction, with revascularisation rates comparable to that of conventional stents," said Giovanni Amoroso, lead author of the study.
TITAN II is a prospective, single-arm clinical trial initiated to further evaluate an enhanced version of the Carillon system, a novel therapy for treating heart failure patients suffering from functional mitral regurgitation.
The Engager transapical valve is comprised of bovine tissue leaflets and a self-expanding nitinol frame designed to facilitate accurate positioning and stability when implanted.
The primary endpoint of the OMEGA trial is nine-month target lesion failure, a composite measure that includes target lesion revascularisation, myocardial infarction and cardiac death in patients with a single coronary artery lesion.
A new JAMA study shows that same-day discharge of selected low-risk Medicare patients who underwent an elective percutaneous coronary intervention (PCI) was not associated with an increased risk of being rehospitalised or having a higher risk of death at two days or at 30 days.
"Results of this study suggest that MitraClip treatment could offer, for the first time, a solution for heart failure patients who have severe mitral regurgitation and are unresponsive to CRT," according to Angelo Auricchio, lead investigator of the study.
"The JenaValve design allows the cardiac surgeon and cardiologist to achieve precise positioning and allows repositioning of the heart valve prosthesis with its unique, patented positioning feelers," according to Friedrich-Wilhelm Mohr, principal investigator of the transapical TAVI system's CE-marking study.
EXCEL is the largest, multicentre, randomised trial to assess the safety and efficacy of the Xience Prime/Xience V everolimus eluting coronary stent systems (Abbott), compared to coronary artery bypass grafting (CABG), or surgery, in select patients with unprotected left main coronary artery disease.
Roberto M Lang, past president of the American Society of Echocardiography, spoke to Cardiovascular News about the use of 3D echocardiography and the future of the technique.
Dean J Kereiakes, USA, comments on the results from the PLATO and TRITON-TIMI 38 trials which randomised patients with acute coronary syndromes to get treatment with either ticagrelor (PLATO) or prasugrel (TRITON-TIMI 38) in comparison to clopidogrel.
Atrium is a leader in medical device technologies for interventional cardiology and radiology, chest trauma care and thoracic drainage, vascular surgery, and general surgery.
The ADVANCE clinical trial is an IDE FDA approved study designed to evaluate the HeartWare Ventricular Assist system as a bridge to heart transplantation for patients with advanced heart failure.
NECTAR-HF is a prospective, randomised, international clinical feasibility study designed to assess preliminary safety and efficacy of chronic vagal nerve stimulation in heart failure patients.
Utilising the heart's natural structures, Carillon (Cardiac Dimensions) is intended to reduce mitral annulus dilatation upon deployment, thereby significantly reducing functional mitral regurgitation.
Blockwise Engineering segmented test stations uniformly radially load a stent or transcatheter heart valve frame with minimal friction to facilitate an accurate measure of the test articles radial resistive force.
The trial for the VMS 3D system will explore this imaging technology in patients with Tetralogy of Fallot or Blue-baby Syndrome.
Nilesh Samani, co-principal investigator of the new study published in PLoS Genetics, said, "The findings provide new insights into and understanding of the causal biological pathways that cause heart disease, and particularly highlight the role of lipids and inflammation."
This is the first catheter combining fiber optic signal acquisition with greater haemodynamic support compared to a standard 40cc intra-aortic balloon catheter.
Run-Lin Gao, described as "the father of interventional cardiology in China" spoke to Cardiovascular News about the Ethica Award recognition, the development of interventional cardiology in China, and the 10th China Interventional Therapeutics (CIT) conference, taking place in Beijing in March 2012.
Martyn Thomas, clinical director of Cardiology and Cardiothoracic Services, St Thomas' Hospital, London, UK, is an enthusiast of transcatheter aortic intervention. In this interview, he told Cardiovascular News why he believes TAVI will be the dominant technology for aortic stenosis within the next 10 years.
The rate of major adverse cardiac and cerebrovascular events is significantly higher with percutaneous coronary intervention in patients with left main or three-vessel disease, according to the SYNTAX trial.
A blinded review of the PLATO trial data found that ticagrelor was associated with fewer complications that directly caused or contributed to death following coronary artery bypass graft surgery.
The self-apposing feature of the Stentys stent ensures optimal apposition during thrombus and vessel spasm relief in the critical initial hours and days after an acute myocardial infarction procedure.
The enCorSQ device (MiCardia) is surgically implanted to treat mitral regurgitation and enables physicians to adjust it without another surgery to correct any recurrent mitral valve regurgitation that may occur due to the progressive nature of the underlying cardiovascular disease.
The recommendation is for first line imaging of the coronary arteries in people with suspected stable coronary artery disease who are difficult to image with earlier generation CT scanners and whose estimated probability of having coronary artery disease is 10-29%.
The Award will help Baker from the University of Glasgow and his team to develop treatments for patients suffering from coronary heart disease and related complications.
The financing will support enrolment into the post CE mark RAMSES trials of Miracor's PICSO technology used during coronary revascularisation procedures following myocardial infarction and other types of acute coronary syndromes to intermittently increase the pressure in the coronary venous system.
The placebo-controlled phase II AMICI trial will evaluate the safety and efficacy of Revascor (Mesoblast), an allogeneic or "off-the-shelf" adult stem cell product used in conjunction with angioplasty and stent procedures to prevent heart failure after a major heart attack.
In the PRODIGY study, 2,000 coronary angioplasty patients were randomised to receive either six or 24 months of dual antiplatelet treatment.
When compared to a bare metal stent, the drug-eluting device demonstrated lower rates of target vessel and lesion revascularisation and stent thrombosis in the EXAMINATION trial.
The highest available recommendation, Class I recognises that prasugrel is considered a "beneficial, useful and effective" treatment option for first-line use in these patients.
New research has found the first evidence that high levels of ionising radiation exposure cause changes at cell level that might represent the body's way of protecting itself against the harmful effects of radiation.
The Barostim neo uses CVRx-patented technology that is designed to trigger the body's own natural blood flow regulation system by electrically activating the baroreceptors to treat high blood pressure.
The annual meeting of the European Society of Cardiology, in Paris, has enhanced the status of TAVI as the "gold standard" for inoperable patients suffering from severe aortic stenosis. However, concerns about high rates of permanent pacemaker implantation after TAVI remain, specialists say.
The panel supports the introduction of intravenous iron – which can be given over a short period of time and is well-tolerated – as a more attractive therapy option to oral iron or erythropoiesis-stimulating agents.
New results from SHIFT (Systolic heart failure treatment with the If inhibitor ivabradine trial), the largest-ever morbi-mortality study of treatments for chronic heart failure, reveal for the first time a direct link between heart rate reduction and quality of life in patients with heart failure.
The 12-week study sponsored by Boehringer Ingelheim will compare three doses of dabigatran etexilate (150mg bid, 220mg bid and 300mg bid) to warfarin in patients with both aortic valve replacements and mitral valve replacements.
Results from the CLARIFY registry show that 44% of the coronary artery disease outpatients had a resting heart rate greater than or equal to 70 bpm, a rate that has previously been shown to be linked with higher prevalence and severity of angina and with more frequent evidence of ischaemia.
Three variants of the updated HeartScore have been formally released at the 2011 ESC Congress in Paris. The new features improve accuracy of the tool.
In preliminary tests from a small trial of 39 patients, computerised tomography (CT) scans called 2nd generation 128 Slice Dual Source "Flash" CT captured quicker images of the entire heart, allowing doctors to better see artery blockages and reduced blood flow through the heart.
The guideline makes a number of new recommendations that are set to improve the way professionals diagnose and treat high blood pressure in in England and Wales.
First used in Europe in 1999 and approved by the FDA in 2006, the occluder is composed of ePTFE patch material supported by a single nitinol wire frame.
Sigmund Silber, of the Kardiologische Klinik Dr Müller, Munich, Germany, discusses the benefits of using the Genous Bio-engineered R stent in patients with atrial fibrillation.
Results from the randomised pilot study showed no significant difference in target vessel failure between Genous and a drug-eluting stent in patients with de novo lesions carrying a high risk for restenosis.
A study led by András Bratincsák from Rady Children's Hospital in San Diego, USA, conducted a review of seven children who underwent intervention between June 2006 and June 2010.
The CoreValve system is the only transcatheter aortic valve available in the world that can treat, without surgery, patients with larger valve openings (up to 29mm).
This bioresorbable vascular scaffold (BVS) can treat a patient's blocked heart vessel and then dissolve within two years, leaving the vessel free of a permanent metallic implant. The treatment of the patient in Japan is part of the ABSORB EXTEND global clinical trial.
Carpentier-Edwards physio tricuspid annuloplasty ring features a three-dimensional waveform shape which allows the ring to conform to the natural shape of a patient's tricuspid annulus during valve closure.
The University of Münster in Germany has successfully bridged its first patient to transplant with the Total Artificial Heart after 89 days of support.
The sex hormone estrogen could help protect women from cardiovascular disease by keeping the body's immune system in check, new research from Queen Mary, University of London, UK, has revealed.
This technology has great potential to save radiation dose during long electrophysiology procedures as the patient does not need to be scanned repeatedly with the angiography system in order to track the catheter.
The system used in cardiovascular angiography, "shortens procedures, decreases the amount of dye injected into patients and reduces radiation exposure for health care professionals and patients," Robert Wilson, ACIST system inventor, states.
Renal stenting with Abbott Vascular's RX Herculink Elite system is safe and significantly reduces blood pressure at nine months, finds the HERCULES study.
The first use of Medtronic’s Symplicity, a minimally invasive, catheter-based procedure for the treatment of resistant hypertension, occurred on 4 August 2011 at Clinica Santa Maria in Santiago, Chile.
Adult human cardiac myocytes have lost their ability to proliferate, perhaps explaining why the human heart cannot regenerate itself, stem cell researchers from the University of California, Los Angeles (UCLA) found.
Data from multiple clinical trials using the OrbusNeich Genous stent were featured during the Healing Symposium at EuroPCR 2011 in Paris, France.
At six months, Acurate TA (Symetis) demonstrated an 87.5% survival rate, a perivalvular leak rate of 0–1 in 97% of the followed patients and functional improvement from baseline in 90% of the cases with a NYHA Class I/II, Joerg Kempfert told delegates at EuroPCR, Paris, France.
For the first time in 50 years, the approval of this oral anticoagulant gives AF patients access to a new treatment for the prevention of AF-related strokes in Europe.
Robert Harrington, Duke University Medical Center, is interviewed on diabetes and multivessel coronary artery disease
Deepak L Bhatt, principal investigator of the SYMPLICITY HTN-3, told Cardiovascular News what the principles behind renal denervation are, outcomes from previous trials and the design of the ongoing SYMPLICITY HTN-3 study.
Ralf Lehmann, Frankfurt, Germany, explains how a combination of clinical and anatomical risk factors may provide the optimal predictive model in patients undergoing multivessel percutaneous coronary intervention.
An 81-year-old woman with severe symptomatic aortic stenosis undergoes TAVI at the Heart and Vascular Institute.
FDA's Advisory Panel voted on 20 July 2011 to recommend approval of the Sapien transcatheter heart valve (Edwards Lifesciences) for the treatment of certain inoperable patients.
Mitroflow (Sorin) contains a proprietary advanced tissue treatment with phospholipid reduction properties which is intended to prevent the potential calcification of the Mitroflow bioprosthetic valve.
The Resolute drug-eluting stent (Medtronic) showed superiority to Taxus drug-eluting stent (Boston Scientific) on the primary endpoint of the RESOLUTE Japan clinical study: in-stent late lumen loss at eight months.
The FDA approval is based upon data from the PLATO study, which shows that Brillinta reduces the rate of a combined endpoint of cardiovascular death, myocardial infarction or stroke compared to clopidogrel.
EP217609 and its specific antidote avidin (Endotis Pharma) were found to be well tolerated in phase I studies. Data were presented at the XXIII Congress of the International Society on Thrombosis and Haemostasis (Kyoto, Japan, 23-28 July 2011).
The Center is the first-of-its-kind collaboration laboratory that will expedite the development of nuclear imaging agents to treat complex diseases like cancer, heart disease and neurological disorders.
Peter Block says renal denervation, which has captured the imagination of interventionalists, has shown to improve glucose metabolism.
Data from high-risk patients requiring early discontinuation of dual antiplatelet therapy due to non-deferrable, non-cardiac surgery show good short and long-term safety outcomes following treatment with the Genous stent (OrbusNeich).
The Ilumien system combines optical coherence tomography and fractional flow reserve technologies on one platform to improve the diagnosis and treatment of coronary artery disease.
The EV1000 clinical monitoring platform (Edwards Lifesciences) displays real-time information about a patient's haemodynamic status simplifying decision-making in the operating room and intensive care unit.
The open-label, multicentre study will evaluate the diagnostic efficacy of flurpiridaz F 18 PET myocardial perfusion imaging (MPI), compared with single photon emission computed tomography (SPECT) MPI in the detection of significant coronary artery disease.
Under the agreement, CryoLife will receive an approximate 19% initial equity ownership in ValveXchange.
CANARY is designed to test the hypothesis that near-infrared guided use of an embolic protection device, during percutaneous coronary intervention can reduce the rate of peri-procedural heart attacks in patients identified as having high-risk lipid core plaques.
BIOFLOW-II is a prospective international trial that will randomise approximately 440 patients with Orsiro (Biotronik) or Xience Prime (Abbott) in a two-to-one manner at 19 European sites.
Results from non-invasive fractional flow reserve combined with computed tomography (FFR-CT) were presented by Bon-Kwon Koo in a Late-Breaking Trials session at EuroPCR.
At one-year follow-up, in-hospital survivors had a mortality rate of 10.5%, whereas 18.4% experienced subsequent major adverse cardiac events.
Coronary bifurcation lesions make up 15% to 20% of PCIs and remain a challenge for cardiologists due to the complexity of the lesions and the increased frequency of associated adverse events, Marcel Beijk writes to Cardiovascular News.
Marvin J Slepian, University of Arizona, USA, discussed the technical and clinical status of the world's only FDA, Health Canada and CE approved total artificial heart at the TechConnect World Summit, Boston, USA, on 14 June 2011.
Based on results observed in the DESSOLVE I trial, Micell has reduced the sample size in its DESSOLVE II CE mark study from 270 to 171 planned subjects.
The National Institute for Health and Clinical Excellence (NICE) has recommended ticagrelor (AstraZeneca) in combination with aspirin as a treatment option in adults with acute coronary syndromes.
NICE published, on 30 June 2011, quality standards for the assessment, diagnosis and clinical management of chronic heart failure in adults, adding to the bank of quality standards already completed.
The European Society of Cardiology (ESC Working Group on Thrombosis) is calling for greater attention to be paid by health care staff to reducing bleeding in patients with acute coronary syndromes undergoing percutaneous coronary interventions, and for increased research in the field.
The first implants have been completed in the InterSEPT trial at the CardioVascular Center at Sankt Katharinen Hospital, Frankfurt, Germany, under principal investigator Horst E Sievert.
In a paper published in the European Journal of Heart Failure, the ESC Working Group on Myocardial Function suggests the use of the general term myocardial remodelling.
Intervention is Viable Option for Low-Risk Patients with Normal Function in Left Ventricle, according to a paper published in Catheterization and Cardiovascular Interventions.
The new stent sizes have a 14mm working length which is 75% longer than the existing Sideguard stents.
Data from the PLATO trial showed that acute coronary syndrome patients, who were intended to follow a non-invasive treatment plan, had fewer events without an increase in bleeding with ticagrelor.
The LIVES study extension showed a relationship between on-treatment serum HDL-C levels and cardiovascular risk, and that pitavastatin provided elevations in HDL-C that continued to increase over five years.
Rapiscan is a new generation pharmacological stress agent for myocardial perfusion scintigraphy in adult patients unable to undergo adequate exercise stress in the diagnosis of coronary artery disease.
"Due to evolving market dynamics in the drug-eluting stent business, we see greater opportunities to benefit patients and grow our business in other areas of the cardiovascular device market," said Seth Fischer, company group chair and worldwide chairman, Cordis.
Drug-eluting coronary stents with a biodegradable polymer significantly improve overall clinical outcomes and reduce stent thrombosis by 50% compared with drug-eluting coronary stents using a durable polymer over a three-year period. These were the findings from a new independent patient-level meta-analysis involving over 4,000 patients, presented at EuroPCR.
Reduction in infarction, improvement in perfusion of the left ventricle and fewer premature ventricular beats were shown in the randomised, placebo-controlled, double-blind safety and feasibility US trial.
The device can be completely resheathed and retrieved before it is released from the delivery system, allowing physicians to reposition the valve at the implant site.
Two-year results of 126 patients from Europe and Canada were presented at the EuroPCR congress in Paris.
A German experience with the device was presented by Martin Bergmann, Department of Cardiology at the Asklepios Klinik St. Georg in Hamburg at the EuroPCR congress in Paris.
JVS-100 is intended to be used in the treatment of patients with heart failure. JVS-100 encodes Stromal-cell Derived Factor 1 (SDF-1), which promotes cardiac repair following a myocardial infarction through activation of natural stem-cell repair pathways.
St. Jude Medical's valve is a clinically-proven replacement for diseased, damaged or malfunctioning aortic heart valves; it has been designed to optimise the blood flow through the valve and has long-term durability.
The system includes the world's first and only cardiac catheter to combine intravascular ultrasound (IVUS) and near-infrared (NIR) spectroscopy to help cardiologists identify and characterise lipid core coronary plaques.
Boston Scientific's system incorporates a unique platinum chromium (PtCr) alloy designed specifically for coronary stenting and is intended to improve the acute performance of coronary stent implantation in the treatment of coronary artery disease.
Eduardo AlegrÃa Barrero, Cardiology and Cardiovascular Surgery, Rotger Clinic, Palma de Mallorca, Spain, and Eduardo AlegrÃa Ezquerra, Cardiology and Cardiovascular Surgery, Guipuzcoa Policlinic, San Sebastian, Spain, explained the first ESC/EACTS guidelines on myocardial revascularisation.
David Smith, principal investigator of the GATEWAY Registry trial, spoke to Cardiovascular News on treatment for acute coronary syndrome patients, their bleeding risk complications from receiving antiplatelet therapy and the GATEWAY Registry that seeks to address this risk.
In patients with aortic stenosis at high risk for surgery, TAVI with the Sapien transcatheter aortic valve (Edwards Lifesciences) was non-inferior to surgical aortic valve replacement for all-cause mortality at one year, 24.2% vs. 26.8%, respectively, according to data presented by Craig R Smith at the American College of Cardiology's Scientific Session in New Orleans, USA.
One-year results from 101 patients enrolled in the second phase of the ABSORB trial evaluating the world's first drug-eluting Bioresorbable Vascular Scaffold (BVS) for the treatment of coronary artery disease were presented at the American College of Cardiology's Scientific Session in New Orleans, USA.
Ralph Brindis, president of the American College of Cardiology (ACC) 2010–2011, spoke to Cardiovascular News about his career, greatest influences, the National Cardiovascular Registry and his efforts as a swimmer for the University of San Francisco Master's Swim team.
Roberto Ferrari, professor of Cardiology at the University of Ferrara, Italy, has recently concluded his term as president of the European Society of Cardiology. He spoke to Cardiovascular News about his current areas of research and his achievements as ESC president.
This is the first randomised clinical trial, in the United States and Canada, to compare a catheter-based mitral valve device to mitral valve surgery. Data from the study were published in The New England Journal of Medicine and presented at the American College of Cardiology's (ACC) 60th Annual Scientific Session.
This system is the world's first-of-its-kind percutaneous minimally invasive treatment option for patients suffering from mitral regurgitation.
Their collaboration will provide clinicians with advanced tools for review of echocardiography and vascular images, and could enhance the speed and accuracy of diagnosis within the non-invasive cardiovascular clinical space.
The new Fetch2 is a manual aspiration device that has the ability to quickly resolve small, fresh thrombus in the coronary arteries.
The FDA cleared station is an automated and portable cardiovascular imaging device which enables physicians, especially primary care physicians and internists, to perform atherosclerosis imaging and cardiovascular risk assessment in their offices, without need for outside referral.
Data from the phase I Co-Rx study suggest that a novel coordinated-delivery tablet of enteric-coated aspirin and immediate-release omeprazole (PA32540) given in conjunction with clopidogrel, dosed at least 10 hours apart, resulted in significantly better inhibition of ADP-induced platelet aggregation when compared to a current standard of care.
Between 2004 and 2006, 697 patients with acute coronary syndromes were enrolled at 37 sites in the United States and Europe after undergoing successful percutaneous coronary intervention. Results of the PROSPECT study were published in The New England Journal of Medicine.
Results of the DECLARE-LONG II trial show triple antiplatelet therapy decreased late lumen loss and restenosis after the implantation of long zotarolimus-eluting stents, resulting in a reduced risk of 12-month target lesion revascularisation compared with dual antiplatelet therapy.
The European Association of Echocardiography (EAE) released a position statement on the use of pocket-size imaging devices in January 2011. Cardiovascular News spoke to Rosa Sicari, one of the authors of the paper published in the European Journal of Echocardiography.
A new study of coronary artery calcium scanning, a simple, noninvasive test that gives patients baseline information about plaque in their coronary arteries has shown that the scan helps them make heart-healthy lifestyle changes and lower their heart disease risk factors.
This new guidance advises that healthcare professionals could consider therapeutic hypothermia as a treatment option for people who are at risk of brain injury after cardiac arrest, under their hospital's usual arrangements for clinical audit/research, governance and consent.
This system incorporates a unique platinum chromium (PtCr) alloy and provides interventional cardiologists a bare-metal stent with improved acute performance in treating patients with coronary artery disease.
Medtronic's CoreValve US pivotal clinical trial will examine an investigational alternative to open heart surgery for patients with severe aortic stenosis.
Flurpiridaz F-18 has the potential to be a new clinical tool for the evaluation of myocardial perfusion that may better evaluate patients with known or suspected coronary artery disease.
Stentys, a developer of stents to treat acute myocardial infarction, will be commercialising its products in Scandinavia, Spain, Switzerland and Poland.
Embrella Cardiovascular is the developer of the Embrella Embolic Deflector System, product designed to reduce the amount of embolic material that may enter the carotid arteries during endovascular procedures.
Azule features OrbusNeich's unique Dual Helix stent design, offering conformability and radial strength on thin, flexible and low-profile cobalt chromium stent struts.
The new heart valve repair ring is designed to restore normal valve function by remodeling the natural valve's shape and size.
The new generation of the Ziehm Vision RFD mobile C-arm offers outstanding imaging and a liquid cooling system which allows it to be used over an almost unlimited period of time, this new version is an attractive choice for hybrid operating rooms (ORs).
Innovative telephone based self care programme for patients with long-term conditions associated with significant reductions in hospital stay and patient care costs.
Two studies recently published have found that a high number of Corevalve implant patients require a permanent pacemaker shortly after the procedure.
A new drug-eluting stent has been designed to increase the antirestenotic performance of the paclitaxel-eluting stent and decrease the risk of stent thrombosis by incorporating cilostazol, according to Seung-Jung Park and colleagues at Asan Medical Center in Seoul, South Korea.
Atritech's Watchman Left Atrial Appendage Closure Technology gives Boston Scientific an innovative product designed for atrial fibrillation patients with high stroke risk.
This transcatheter valve enables clinicians to replace failing aortic valves with a minimally invasive procedure.
Boston Scientific's third-generation bare metal stent offers improved acute performance in treating patients with coronary artery disease.
The Neovasc Reducer, COSIRA's device under study, is designed to establish a permanent and controlled narrowing of the coronary sinus.
Medtronic will fund and provide its drug-device expertise as Nile executes on its phase I clinical trial to assess the pharmacokinetics and phamacodynamics of cenderitide delivered through Medtronic diabetes pump technology.
This intravascular ultrasound catherer offers improved deliverability and additional radiopaque markers to accurately perform angiographic lesion length assessments.
This is the first document offering guidance regarding core measures for quality and assessment in percutaneous coronary intervention programmes.
This balloon catheter is designed to penetrate severely narrowed lesions and tortuous anatomies.
The study revealed freedom from structural valve deterioration in 93.3% of aortic valve replacement patients and in 95.3% of mitral valve replacement patients.
Target lesion revascularisation was 5.7% and stent thrombosis was 1.1%, these were some of the results published in the February issue of EuroIntervention.
This clinical investigation is being conducted to support CE mark approval of the company's MiStent drug-eluting coronary stent (DES) system.
India is one of the fastest-growing drug-eluting stent markets in the world, with the number of coronary drug-eluting stents implanted annually estimated at more than 150,000. In 2010, the company received approval from the Drugs Controller General of India to market these systems.
The Registry shows that patients receiving the CoreValve system (Medtronic) have high rates of procedural success (98%), low rates of adverse events (2.1%) and substantial improvements of cardiac function. The study results were published online in the journal Circulation.
Experts tell Cardiovascular News why it is too early to conclude what the role of absorbable devices will be and explain what still needs to be investigated.
Olaf Wendler and Martyn Thomas compare the results of the PARTNER randomised trial with those of the SOURCE European registry and speculate on reasons for differences in outcomes.
GATEWAY registry will assess the performance of the device in providing anti-ischaemic protection while providing bleeding safety by the use of shortened dual antiplatelet therapy.
PeriPatch-BV (Neovasc) products are used in a variety of general surgical and cardiovascular applications and as components in implantable heart valves and other medical devices.
The aim of this paper is to provide an overview of the true risk of radiation exposure in pregnant practitioners who perform cardiac catheterisation procedures and best practice guidance to reduce radiation exposure.
The FDA also informed that rosiglitazone's labels advice that the drug should only be used in patients already being treated with these medicines and in patients whose blood sugar cannot be controlled with other anti-diabetic medicines.
The acquisition price of €3.15 million (US$4.3 million) is paid from existing cash balance.
The Ingenuity TF PET/MR (Philips) is used to screen patients at high-risk for heart disease to ultimately treat diseased cells prior to the formation of dangerous coronary plaques.
The clinical endpoint of the trial is the ability to treat the patient without the incidence of major adverse cardiovascular events, also to monitor the radiation exposure of interventional cardiologist while using the CorPath (Corindus) technology.
PLATINUM China clinical trial is designed to evaluate the safety and effectiveness of the Element stent in the treatment of patients with a single de novo atherosclerotic lesion.
Pieter Kappetein explains how the SURTAVI trial will use the same SYNTAX trial concept, with a heart team deciding which patients can be randomised to transcatheter aortic valve implantation (TAVI) with the CoreValve device or to surgical aortic valve replacement.
Data generated on the Xience V drug-eluting stent from the DESTINY trial supported the CE marking. Marc Bosiers, principal investigator of the trial, concluded that the use of everolimus-eluting stents can potentially provide significantly better outcomes for patients with critical limb ischaemia in the lower leg.
In the study, the major adverse cardiac events free survival rate in patients treated with the Genous stent, from OrbusNeich, was 80.6% after one year.
Khaldoun G. Tarakji and colleagues, researches of the study, concluded that occurrence of stroke among patients undergoing coronary artery bypass graft has decreased over the last 3 decades despite an increasing patient risk profile.
The paper titled "Radiation safety program for the cardiac catheterization laboratory" includes recommendations designed to enhance patient and staff safety in cardiac procedures involving radiation.
The Synergy drug-eluting stent is designed to provide the same degree of restenosis reduction as a conventional drug-eluting stent while offering faster and more complete vessel healing after stent implantation.
The study published in The New England Journal of medicine also showed that differences in underlying tissue type can change the risk profile of a lesion.
The CoreValve system is designed to provide a minimally invasive, non-surgical treatment option for patients with symptomatic, severe aortic stenosis who are at high risk, or are ineligible, for open-heart surgery.
The new intracardiac ultrasound and intracardiac echocardiography (ICE) system offers real-time image guidance, visualisation of cardiac anatomy and great portability.
A multicenter, randomised controlled trial conducted from February 2003 to February 2009 among 757 participants undergoing first-time elective coronary artery bypass grafting showed insignificant differences in the use of radial artery and saphenous vein grafts.
The next generation device offers physicians smoother device deployment while maintaining all the benefits of the original Mynx.
The Absorb device, from Abbott, can treat blocked coronary arteries and then dissolve, leaving the patient's vessel free of a permanent metallic implant.
Boston Scientific paid US$193 million to acquire the remaining 86% of Sadra Medical's equity.
The stent is built on an ultra-low strut thickness (65µm) cobalt chromium stent platform, using an intelligent hybrid of close and open cells allowing for morphology mediated expansion.
The ADVANCE trial demonstrated that 94% of the investigational device patients enrolled in the study achieved a survival endpoint at 180 days with the device.
Indulis Kumsars, chief of the catheterisation laboratory at Pauls Stradins Clinical University Hospital in Riga, Latvia, enrolled the first patient.
The new device is designed to improve acute coronary syndrome revascularisation following primary percutaneous coronary intervention.
The American Heart Journal reported on 21 December 2010 results of a clinical trial evaluating AMR-001 for the treatment of damaged heart muscle following acute myocardial infarction.
The first CoreValve transcatheter aortic valve implantation (TAVI) procedure was performed by David H Adams, and Samin K Sharma, at The Mount Sinai Medical Center in New York City.
Next-generation pericardial tissue valve provides physicians with an advanced valve, designed for superior hemodynamic performance.
Simple blood tests with new high sensitive assay show that even small changes accurately forecast 90-day prognosis.
The APPOSITION III study will allow physicians to use the Stentys self-apposing stent in a real-life setting for the treatment of acute myocardial infarction patients.
Approval is expected to expand access to care for patients with severe aortic stenosis whose femoral arteries are too narrow or are compromised due to disease.
Brilique (AstraZeneca) has been authorised for the prevention of atherothrombotic events in adult patients with acute coronary syndromes.
Martin B Leon, professor of Medicine, founder and chairman emeritus of the Cardiovascular Research Foundation, will serve as principal investigator of the study.
Abiomed has received conditional approval from the FDA to begin MINI-AMI, a randomised trial to assess the potential role of the Impella 2.5 in reducing infarct size in patients with STEMI.
Novel approach to angioplasty uses standard guiding catheters; promotes widespread adoption of transradial approach in the USA.
The surprisingly good results from the inoperable PARTNER cohort provides confidence that additional TAVI studies are justified in lower risk aortic stenosis patients and in other selected patient cohorts, Martin Leon said.
The SeQuent Please device should be considered for patients with in-stent restenosis and in cases where there are clinical reasons to minimise how long anticoagulant therapy is used.
Results of the EMPHASIS-HF study have shown that rates of heart failure hospitalisation, all-cause hospitalisation and all-cause mortality were also significantly reduced.
"We will witness the introduction of new devices to enrich the treatment portfolio, the development of image guidance to plan and execute the procedures, and the evolution of new clinical trends and, potentially, updated guidelines," Franscesco Maisano, Milan, Italy, writes to Cardiovascular News.
Innovative percutaneous aortic valve replacement system provides entry into emerging market of structural heart disease.
Tricuspid Annuloplasty ring is designed to adapt to the heart's natural tricuspid valve
Blue Medical will introduce the CoCr stent on DEB, called Pioneer and its DEB, called Protege across Europe by the end of the year.
Data from the EURIKA study highlight the importance of identifying and managing patients with uncontrolled cardiovascular risk factors early.
Carpentier-Edwards biological heart valves were developed by Carmat co-founder and CSO Alain Carpentier and will be, for the very first time, used in an artificial heart.
George Dangas, co-director of Transcatheter Cardiovascular Therapeutics (TCT) conference, spoke to Cardiovascular News about his career, research and challenges facing interventional cardiology today.
Positive nine-month results from the first 45 patients enrolled in the second stage of the ABSORB trial were presented at the 22nd annual TCT conference in Washington, DC.
Balloon-expandable TAVI should be the new standard of care for patients with aortic stenosis who are not suitable candidates for surgery, Martin Leon told delegates at the TCT conference.
SPIRIT IV and SORT OUT IV trials compared everolimus-eluting stents to paclitaxel- and sirolimus-eluting devices in large groups of patients.
Results of the randomised, multicentre PERFECT STENT study were presented by Jochen Wohrle, Ulm, Germany, at the TCT conference.
Medtronic has received FDA approval for the Integrity coronary stent system. The device has been shown in bench testing and in blinded in vivo physician assessment studies to be highly deliverable.
PROMUS CE mark approval is well received by physicians.
Magna Mitral Ease valve designed to enhance implantation in the challenging mitral position.
The effects on the level of platelet aggregation achieved after switching from Plavix 75 mg once daily maintenance dosing plus aspirin to Effient 10 mg once daily maintenance dosing in patients with acute coronary syndrome.
Technology advances allows physicians to achieve enhanced control and accuracy in the deployment of the CoreValve device.
On the first day of the 24th EACTS meeting, in Geneva, Cardiovascular News interviewed Michael Mack and Thomas Walter on TAVI trends in Europe and the USA.
InfraReDx, Inc has announced clearance from the US FDA to market LipiScan IVUS, thus being the first catheter to combine near-infrared spectroscopy with intravascular ultrasound a valuable new tool to characterise coronary plaque and improve coronary stenting.
Additional analysis of 18 month data of the SORT OUT III trial provides detail on safety and efficacy outcomes in patients with diabetes, acute coronary syndrome, or treatment of multiple lesions.
The FDA has approved an Investigational device exemption supplement that allows HeartWare to enroll a second allotment of 54 patients in its ADVANCE bridge-to-transplant clinical trial, which is designed to evaluate the HeartWare® Ventricular Assist System for patients with end-stage heart failure.
Cortisol levels in hair may be the first biomarker to measure chronic stress, which is linked to a higher risk of having a heart attack.
Large-scale study shows positive results for drug ivabradine in reducing chronic heart failure; EINSTEIN-DVT also shows positive outcomes for rivaroxaban.
Publication of decade follow-up test results on first CYPHER sirolimus-eluting coronary stent patient reveal the stent's outstanding efficacy and safety.
"Superior deliverability" distinguishes new medical drug-eluting stent for coronary artery disease from rivals, according to the company.
Update on chronic heart failure replaces July 2003 clinical guideline.
Conclusion comes as the result of an unprecedented study into the association between system delay and outcome in patients with STEMI treated with primary PCI.
School of Medicine becomes first institution in the world to use multi-colour CT technology in a pre-clinical setting.
Clinical evaluation of a revolutionary new device technology for the treatment of vascular disease has begun.
Target Lesion Failure rates marginally lower for Resolute, according to journal.
Committee voted 7 to 1 in favour of Astrazeneca drug which outperforms rival plavix according to PLATO study.
Evolve trial to evaluate company’s fourth-generation Synergy drug-eluting coronary stent.
Rivaroxaban shows superiority to standard therapy in net clinical benefit; full data set to be presented at ESC 2010 Hot Line Session.
New device optimises harvesting technique to improve graft quality for coronary artery bypass graft patients.
Lancet study may send out the wrong signals about cholesterol levels, ESC warns.
Methodist to be first Texan hospital to offer FDA-Approved total heart replacement for transplant patients.
New JAMA study strengthens argument against the medication's use on type 2 diabetes patients.
Authors' report published in JAMA June 2010 issue suggests patient outcomes "have not improved in all areas".
Company plans to advance development of the drug following results presented at the American Diabetes Association's 70th Scientific Sessions.
Data presented at 20th ESH meeting shows a relative risk reduction of nearly a third after 12 months of treatment.
Results suggest transcatheter valve implantation is best treatment choice for patients undergoing non-conventional surgery.
Six-month data shows positive results for revolutionary BVS technology.
Twelve-month data shows the OrbusNeich Genous Bio-engineered R stent to be effective in diabetic patients.
Dr David Smith advocates a shift away from stents that involve the need for dual-antiplatelet therapy.
"Europe has continued to be the stage upon which major developments with this technology have debuted," Dr Martyn Thomas claims.
The Taxus Element Stent System is the company's third-generation drug-eluting stent (DES) technology; it received CE mark approval in May.
Josep Brugada speaks to Cardiovascular News about his career, his proudest moments and his association with Barcelona FC.
Five studies presented at Society of Cardiovascular Computed Tomography Annual Scientific Meeting held in Las Vegas.
New national report defeats the myth that airline passengers with heart conditions pose a safety risk.
Gore reaffirms commitment to multinational, randomised study on PFO closure in stroke.
Acquisition makes St Jude Medical the first company to offer both optical coherence tomography and Fractional Flow Reserve technology.
Hannu Romppanen and Matti Niemelä discuss the use of the Promus Element everolimus-eluting stent system
A study presented at ACC 2010 has shown that lighter control of heart rate can be as effective as strict control
Francesco Prati discusses how drug-eluting stents have been found to dramatically reduce the incidence of restenosis.
OrbusNeich lead the way in stent research with AICT showcase.
Peter Damman, Academic Medical Center, Amsterdam, The Netherlands, presented the subset analysis from 1,236 diabetic patients at EuroPCR.
The analysis will evaluate the cost effectiveness of an FFR-guided intervention strategy for patients with multivessel coronary artery disease in eight countries.
Randomised study of nearly 3,000 patients will evaluate Company's third-generation drug-eluting stent technology.
Robert-Jan Van Geuns and Maciej Lesiak presented studies showing that the Tryton Stent demonstrated procedural rates of 94% and 98.5% respectively.
One-year clinical results from innovative RESOLUTE All Comers Trial show excellent performance for Resolute DES in challenging patient population.
Positive six-month results from the first 45 patients enrolled in the second stage of the ABSORB trial were presented at the EuroPCR meeting in Paris.
Transcatheter aortic valve delivers positive clinical outcomes through two years in major international study and achieves 10,000-patient milestone.
Martyn Thomas reported at EuroPCR that the device demonstrated promising one-year mortality rates for high-risk patients, many of whom were too sick to undergo open-heart surgery.
Nevo, incorporating RES Technology, continues to support excellent safety and efficacy outcomes vs. the Taxus Liberté according to data presented at EuroPCR 2010.
In the XIENCE V USA study, the device demonstrated low blood clot rate at one year; in SPIRIT V Diabetes Trial, Xience V had zero cases of stent thrombosis at one year.
Preliminary results also show MitraClip device maintains durability through two years in patients treated for mitral regurgitation.
Biotronik's first drug eluting balloon is indicated for the treatment of coronary in-stent restenosis.
The Company plans to launch the product in European markets this week and in the US next month.
A new version of a web"based platform designed to share emergent patient information between Emergency Medical Services (EMS) and hospital care teams has been launched.
A new side-branch stent for the treatment of bifurcation lesions has been launched by Tryton Medical. The Tryton Side Branch Stent System is now available in Austria, Belgium, Denmark, Ireland, Luxembourg, the Netherlands, Norway, Poland, Portugal, Spain, and the United Kingdom. Bifurcation lesions are the largest unmet need confronting interventional cardiology.
Prof D. Carrié speaks to Cardiovascular News about progress in percutaneous coronary intervention.
Results from the SORT OUT III trial presented at ACC 2010 surprised delegates by showing that the Cypher stent was associated with significantly lower rates of major adverse cardiac events (MACE), all-cause mortality, myocardial infarction (MI) and target lesion revascularisation (TLR).
A new device designed to combat mitral regurgitation (MR) met primary safety and efficacy endpoints in the Endovascular Valve Edge-to-Edge Repair Study (EVEREST) II, it was reported during a late-breaking data session at the American College of Cardiology 2010 scientific sessions in Atlanta, USA on 14 March.
Results from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial showed that a combination of fenofibrate and simvastatin failed to reduce risk of fatal cardiovascular events compared to simvastatin alone.
Clinical data presented at the Joint Interventional Meeting (JIM) 2010 in Rome provide further evidence that Orbus Neich's Genous stent could be a treatment of choice for patients not suitable for long-term dual antiplatelet therapy.
Alfredo R Galassi, director of diagnostic and cardiovascular interventional laboratory, Ferrarotto Hospital University of Catania, Italy, and associate professor of cardiology, University of Catania, Italy, tells Cardiovascular News International about his career journey to date, and his love of sailing
The US Food and Drug Administration has approved Thoratec's PMA (pre-market approval) supplement, allowing the use of its HeartMate II left ventricular assist system for destination therapy.
Three-year results of the RESOLUTE trial include no new target lesion revascularisations and no instances of stent thrombosis.
Alain Cribier, who performed the first double balloon technique for dilatation of valvular stenosis in 1985, spoke to Cardiovascular News International about the evolution of percutaneous techniques.
Since the first percutaneous implantation of a pulmonary valve in a human was reported for the first time in 2000, transcatheter heart valve procedures have recorded constant double-digit growth year-on-year, offering a new option to candidates not suitable for conventional surgery.
The everolimus-eluting stent, Xience V has been approved by the Japanese Ministry of Health, Labour and Welfare (MHLW), Abbott has announced.
Carlo Briguori, Naples, Italy, discusses the difficulties in compliance and low response to dual antiplatelet therapy.
The results of the ARBITER 6-HALTS trial raised questions and prompted heated
discussion at the American Heart Association scientific sessions.
New guidance recommending the use of prasugrel in combination with aspirin as an option for preventing blood clots in the arteries of people with acute coronary syndromes has been published by the National Institute for Health and Clinical Excellence (NICE).
The latest progress in traditional heart surgery, minimally invasive and transcatheter techniques were presented in a two-day congress at the Heart Centre, University of Leipzig, Germany.
OrbusNeich have announced the initiation of patient enrolment in the randomised clinical trial of the Combo Bio-engineered Sirolimus Eluting Stent (Combo Stent).
The transfemoral and transapical valve implantations were performed by Osaka University in preparation for application submission to the Japanese Ministry of Health, Labour and Welfare.
Expanded indications for Abbott's coronary stents are based on data from the SPIRIT family of randomised trials.
Results for the Carpentier-Edwards Perimount Magna Ease valve were presented at the European Association for Cardio-Thoracic Surgery annual meeting, in Vienna, Austria.
Percutaneous heart valves get a boost as Abbott acquires Evalve.
The results of the PROSPECT trial show that the identification of lesions can be enhanced by intravascular ultrasound.
Abbott Vascular’s Xience V stent has been shown to be superior to other drug-eluting devices in two trials reported at the Transcatheter Cardiovascular Therapeutics 2009 annual meeting in San Francisco, California, USA.
Two-year follow-up data from the HORIZONS-AMI trial presented at the TCT conference show that Taxus and bivalirudin maintained their advantage in ST segment elevation myocardial infarction patients that was shown at one year.
Two-year results of the LEADERS trial show an increasing trend towards a safety benefit for the biolimus A9-eluting stent with abluminal biodegradable polymer.
Two-year results of the LEADERS trial show an increasing trend towards a safety benefit for the biolimus A9-eluting stent with abluminal biodegradable polymer
Trial shows an FFR-guided strategy to treat multivessel coronary artery disease both improves outcomes and is cost-saving compared to procedures using angiography alone
Pre-clinical data were presented at the TCT conference. OrbusNeich set to initiate patient enrolment in human clinical trial in fourth quarter.
Minimised ExtraCorporeal Circulation (MECC) system offers new approach to improve patient safety during heart surgery.
Data supporting the long-term safety and effectiveness of renal sympathetic denervation in treating patients with chronic hypertension were presented at the TCT conference.
Three-year Taxus ATLAS results show safety and efficacy advantages for Boston Scientific Taxus Liberte Stent.
Data on histopathology, mechanical integrity of the device, left ventricular reverse remodeling, and mitral valve annulus dimensions were released in San Francisco.
Mega has a smaller insertion point than traditional 50cc intra-aortic balloons and offers additional blood volume displacement for intra-aortic balloon counterpulsation.
Three-year results of the Medtronic's RESOLUTE trial include no new target lesion revascularisations and no instances of stent thrombosis.
New diagnostic tool combines St Jude Medical's PressureWire Aeris with GE's Mac-Lab Hemodynamic Recording System.
Latest findings from ENDEAVOR IV clinical trial also showed 91% reduction in risk of very late stent-related blood clots for Endeavor patients.
Boston Scientific announced on 21 September 2009 release of its CE-marked, next-generation iLab System Software for the iLab Ultrasound Imaging System.
PLATINUM clinical programme will support US and Japanese approval of new everolimus-eluting coronary stent system.
Valturna combines in a single pill valsartan, an angiotensin receptor blocker, with aliskiren, the only approved direct renin inhibitor.
Implantation of the GDS Accucinch System significantly reduced the patient's mitral regurgitation without requiring open heart surgery.
OrbusNeich's Genous Bio-engineered R stent is feasible and safe in patients who need coronary revascularisation before undeferrable non-cardiac surgery and have to discontinue dual antiplatelet therapy.
The addition of the angiotensin receptor blocker (ARB) Diovan to a non-ARB-based treatment regimen for high blood pressure provided a 45% relative risk reduction in cardiovascular events.
Company plans fourth-quarter launch for XIENCE V in China, the second-largest drug-eluting stent market in the Asia-Pacific region.
Acquisition expands vascular portfolio and provides Abbott with global leadership position in non-surgical treatment option for mitral valve repair.
According to the company, Avantgarde represents the latest and most advanced drug-free technology for the treatment of cardiovascular disease.
The procedures with the new lower-profile Ascendra 2 transapical delivery system were conducted at the University of Leipzig Heart Centre, Germany.
Kornelia Kotseva, Imperial College, London, UK, analyses the results of the EUROASPIRE surveys.
Results of the PLATO, RE-LY, PRAGUE-7, and PROTECT trials were presented during the conference in Barcelona, Spain.
Andre Rubboli, Bologna, Italy, analyses a polymer-free, Carbofilm coated stent eluting tacrolimus from abluminal reservoirs, which has been widely tested in patients with either stable or unstable coronary artery disease.
Study found that women may have a slightly higher risk following an acute coronary syndrome, but that these differences appear to be attributable to severity and type of syndrome.
TYPHOON and STRATEGY, two trials with long-term follow-up periods, showed sustained benefits for the Cypher stent when compared to bare metal stents.
The clinical program for Xience V studied patients in the US, Canada, Europe, South Africa and Asia-Pacific, and demonstrated excellent long-term results.
Patients with a certain genetic variation who received clopidogrel had a decreased platelet response to treatment according to a new study.
Study showed that, from 1995-2006, hospital 30-day death rates decreased significantly for Medicare patients hospitalised for a heart attack.
Chinese scientists presented one of the first studies suggesting that oxycholesterol may be the most serious cardiovascular health threat of all.
A website dedicated to the use of the Syntax Score, a novel angiographic tool used to measure the complexity of coronary artery disease, was launched at EuroPCR.
In the randomised NEVO RES-I trial, the Nevo sirolimus-eluting stent had significantly lower in-stent late lumen loss than the Taxus-Liberté device.
The systolic blood pressure goal of ?140mmHg was met by half of patients receiving the drug, according to a study presented at the American Society of Hypertension meeting.
Jamie S Sutherell, cardiology fellow at Cincinnati’s Children Hospital, US, analysis the lack of paediatric medical devices and off-label use.
Silvia G Priori spoke to Cardiovascular News about her career, research, joy in spending time with her two children, and the occasion she took beta-blockers before meeting Dr Mike Rosen.
At 38mm, it is the longest available drug-eluting stent and affords a more efficient treatment option for the estimated 8-10% of patients with long lesions.
Daiichi Sankyo and Lilly' Effient reduces thrombotic cardiovascular events in patients with acute coronary syndromes managed with percutaneous coronary intervention.
The number of PTCAs performed between January and March in Western Europe was 0.3% higher than in the Q4 2008 and 1.2% lower than in Q1 2008. The DES penetration rate was measured at 51.9%.
Study will assess Tryton side-branch stent in real-world settings. The device demonstrated excellent six-month clinical results in a first-in-man study of the system in 30 patients.
Approved to reduce the risk of cardiovascular hospitalization, Multaq will be launched this summer in the US by Sanofi-aventis.
The dataset demonstrated excellent results in high-risk patients with complex coronary lesions, including high procedural success rate and sustained safety.
Study shows that phone and Internet-based interventions make rehabilitation programmes more accessible after a heart attack.
Results from the largest and longest examination of enzyme replacement therapy in female demonstrated benefits on kidney function, heart function and burden of disease.
Investigation will study the performance of the Xience Prime everolimus-eluting stent, currently an investigational device, for the treatment of coronary artery disease.
Study presented at the XV International Symposium on Atherosclerosis shows novel synthetic statin is safe and well tolerated in combination with warfarin.
Trilipix delayed-release capsules in combination with rosuvastatin calcium achieved individual and combined lipid targets in patients with mixed dyslipidemia and type 2 diabetes.
The Society for Cardiovascular Angiography and Interventions encourages healthcare providers to apply the findings of BARI-2D, when appropriate, to their patients, as studied in this trial.
Analyses conducted from Pre-RELAX-AHF study showed that drug for acute heart failure treatment was the strongest independent predictor of improved longer-term outcomes.
Boston Scientific has received approval to market its new drug-eluting stent specifically designed for treating small coronary vessels.
Study analyses the outcomes of 47,967 patients entered into the Swedish Coronary Angiography and Angioplasty Registry between 2003 and 2006.
How much work cardiologists will take away from surgeons was one of the topics debated at the ESCVS meeting held in Warsaw, Poland, from 30 April–2 May.
BioInvent International AB has announced that its therapeutic antibody product candidate has reached the primary endpoint in its phase I trial, and was safe and well tolerated.
Research led by University of Calgary, Canada, shows ethnic variation in heart disease symptoms.
Analysis of data from 162 centres worldwide between 1995 and 2006 showed that tamponade caused the highest number of fatalities, followed by stroke and atrioesophageal fistula.
Substudy from the TRITON-TIMI 38 trial showed that patients who took prasugrel and had a reduced function of the CYP2C19 gene did not have an increased risk of cardiovascular events.
Dr Dirk J van Veldhuisen analyses the findings on the use of CRT for heart failure patients classes I and II and how the results will impact in future studies.
CID has announced that it has successfully defended European Patent 1277449 against an opposition filed by Conor Medsystem on proprietary drug-eluting stent technology.
Study showed that material commonly used in the production of such medical plastic devices as intravenous bags and catheters can impair heart function in rats.
Innovations in decision support, quality CPR and information management solutions help reduce critical time to lifesaving treatment.
Dr Maciej Banach, Medical University of Lodz, Poland, presented at The European Society of Cardiovascular Surgery meeting a range of studies showing the benefits of statin.
Results of a Chinese study showed that patients receiving drug-eluting stents had considerably higher three-year rates of target vessel revascularisation.
Study found that mice that lack the receptor for one type of the fatty compound prostaglandin have lower blood pressure and less atherosclerosis than their nonmutant brethren.
Software designed for the interpretation of coronary CT angiography helps radiologists determine which cases are high priority and should be read first.
OrbusNeich bio-engineered R stent showed low rate of stent thrombosis in studies presented at the Angioplasty Summit TCT Asia Pacific 2009.
Cardiovascular News talks to Professor Christian Hamm, Bad Nauheim, Germany, about his influences, current areas of research, and the challenges facing cardiology.
Eating fatty fish and marine omega-3 fatty acids, which are found in fish oil, seems to protect men, according to one of the largest studies to investigate the association.
The penetration rate was measured at 51.6% in Western Europe: this represents the fifth consecutive increase since the third quarter 2007.
Healthy individuals may be able to cut their risk of cardiovascular disease by 50 to 60% by taking a single pill that combines three blood pressure lowering drugs, a statin, and aspirin.
The benefits of rosuvastatin extend to preventing venous thromboembolism, according to analysis of the JUPITER study.
Professor Alfredo Galassi, Catania, Italy, analyses the techniques for the treatment of bifurcation lesions.
AnalyST Accel and Current Accel provide new alerts and insight into cardiac conditions such as atrial fibrillation and ischaemia.
Using clinical variables, American researchers created a method to estimate bleeding risk and help lessen the chances that heart attack patients will experience this complication.
Tea consumption simultaneously increased blood vessel reactivity and reduced both blood pressure and arterial stiffness.
The treatment of mitral regurgitation using the MitraClip system has demonstrated success in the first UK patients who have undergone the procedure.
Study shows screening did not result in a significant reduction in the rate of heart attacks or cardiac death compared to patients who were not screened.
The Design Zentrum Nordrhein-Westfalen has bestowed the red dot design award on the world's smallest cardiopulmonary support system Cardiohelp.
Aerobic exercise training appears safe for patients with heart failure and was associated with a modest reduction in the risk of death and hospitalisation.
Cognitive behavior therapy and supportive stress management appear more effective than usual care for treating depression after coronary artery bypass surgery.
Dr Erica Spatz tells how a new analysis estimated the number of older American adults who may benefit from statin therapy following the JUPITER trial results.
Dr Alfredo Galassi, Italy, launches practical handbook for interventional cardiologists willing to improve skills in the field of revascularisation of CTO.
Study showed that people in heart failure have less of the enzyme needed to activate a hormone that helps reduce extra sodium and fluid that tax the cardiovascular system.
The new system combines the Driver bare metal stent, platform of the company's coronary stents, with an improved delivery system.
A new report from the trial shows that the advantages of bypass surgery are less obvious once quality of life and economic data are included in the analysis.
The updated document includes an entirely new section on managing patients who are hospitalised with acute heart failure.
Study shows that the procedure not only failed to reduce deaths and hospitalisations in heart failure patients but also did not improve quality of life.
New analysis from the trial show that rosuvastatin significantly cut the risk of venous thromboembolism by 43% compared to placebo.
Comparative effectiveness study of 262,700 American patients was presented at the American College of Cardiology's 58th Annual Scientific Session in Orlando, USA.
Noninvasive sensor test is "highly predictive" of heart attack or stroke for people who are considered at low or moderate risk.
Standard and Poor’s has raised the company’s rating outlook to positive from negative, while affirming the company’s corporate credit rating at BB+.
New data from the American Centers for Disease Control and Prevention provide additional evidence that limiting amount of sodium can prevent and reduce hypertension.
Large cardiovascular outcomes study to be initiated will evaluate the effects of the drug once weekly compared to standard of care with traditional antidiabetes medications.
Study found that progression of a rare type of cardiomyopathy linked to a genetic mutation may be rapid and often results in early death.
This year's Artery Explorer tour kicks off this month, urging people across the USA to take action against atherosclerosis.
The hsCRP test used in the JUPITER trial will be available in the company's booth in the upcoming event in Orlando, USA.
Company will launch a complete image management system that provides image management software and image acquisition, storage, display and web distribution devices.
Dr Ascan Warnholtz, University of Mainz, Germany, puts into perspective the results of the trial and the use of myocardial fractional flow reserve.
Results of a study performed to assess the clinical and angiographic long-term outcome after implanting drug-eluting stents were published in JACC Cardiovascular Interventions.
Abbott has announced the initiation of the next phase of the ABSORB trial to evaluate the safety and performance of the fully bioabsorbable drug eluting coronary stent.
Document helps physicians deciding whether it is appropriate to treat chest pain with medical therapy alone or prescribe medical therapy and also perform revascularisation.
The aim of the campaign is to improve quality of care and reduce mortality rates among acute coronary patients.
According to study published in the Journal of the American College of Cardiology, both stents provide comparable clinical and angiographic outcomes.
The SOS study showed that Taxus reduced restenosis, target lesion revascularisation, and target vessel failure compared with bare metal stents.
St Jude Medical has announced European CE mark approval of its Genesis neurostimulation system for managing chronic angina.
Catania coronary stent with NanoThin Polyzene-F shows sustained 12-month benefit in the treatment of de novo lesions.
Cordis has announced plans to launch NEVO II, a global, head-to-head clinical trial to compare the Nevo sirolimus-eluting coronary stent to the Abbott's Xience V everolimus-eluting stent.
0
Survey shows that large proportions of European coronary patients are failing to achieve lifestyle, risk factor and therapeutic targets for the prevention of further disease.
Data in The Lancet demonstrate Abbott's device is absorbed within two years, leaving behind blood arteries that appear to move and function similar to untreated vessels.
BridgePoint Medical has completed the first human clinical case using its CrossBoss CTO Catheter and Stingray CTO Re-Entry System.
Report proposes an explanation for the apparent paradox of improved prevention, falling mortality rates but stable rates of hospitalisation.
Results of the trial were published in the Journal of the American College of Cardiology showed device is easy to use and provides excellent hemodynamic support.
The Voyager NC is a balloon dilatation catheter designed to optimise the treatment of patients with coronary artery disease during angioplasty procedures.
Following an acute coronary syndrome, patients who receive a medication to reduce the risk of gastrointestinal bleeding that may be associated with the use of clopidogrel and aspirin have an increased risk of hospitalisation or death.
Patients with abnormal diastolic function in the left ventricle have a substantially lower maximum capacity for exercise, according to a study published in the JAMA.
An examination of clinical practice guidelines for treating cardiovascular disease finds that current recommendations are largely based on lower levels of evidence or expert opinion.
In the not-too-distant future, a simple blood test may predict the genetic risk of suffering a heart attack, according to research from the McMaster University, Canada.
New developments for prevention/reduction of atherothrombotic events in patients with acute coronary syndromes who undergo PCI were presented at the VEITHsymposium 2008.
Study shows that some patients who received beta-blockers before and around the time of non-cardiac surgery appear to have higher rates of infarction and death within 30 days of the procedure.
Many among the abstracts at the AHA Scientific Sessions 2008 were focused on the setting of ACS. Two such sessions were included among the high-profile Late-Breaking Clinical Trials. A third examined treatment for children with familial hypercholesterolemia.
Cardiovascular News talks to Dr Kim Fox, immediate past-president of the European Society of Cardiology (ESC), about getting into cardiology, his interest in ischaemic heart disease and the future of cardiovascular medicine.
0
0
0
0
0
The European Society for Cardiology Committee for Practice Guidelines has released three new sets of updated guidelines in these three areas of cardiovascular care.
Two studies showed that statin treatment with Crestor had no effect in patients with chronic heart failure, while a one-a-day capsule of omega-3 can reduce mortality and admission to hospital for cardiovascular reasons.
Results from a post-hoc analysis of the data from the trial show that dronedarone reduces stroke risk.
The TIME-CHF trial showed that the use of natriuretic-peptide levels rather than symptoms alone to guide the medical management of heart failure failed to influence the primary endpoint of hospitalisation-free survival over 18 months.
Study was designed to investigate how the standards set in 2003's Third Joint European Task Force guidelines on cardiovascular disease prevention have been put into practice.
Results from a new study suggest that patients' brains may retain the capacity to rewire through a treadmill exercise programme months or years after conventional physical therapy has ended.
Results from a Canadian study show that only 40% of ischaemic stroke patients who had atrial fibrillation had received warfarin before their stroke.
A project involving institutions from six European countries, euHeart has been created to improve the diagnosis, therapy and treatment of cardiovascular disease.
A new review of clinical trials, published online in Nature in August, revealed that the number of registered cardiology studies declined during a recent two-year period.
Study over the first five years of life found that there is a link between the pace of weight gain in early childhood and high blood pressure in adulthood.
Among key sessions at 2008 American Society of Hypertension (ASH) Annual Meeting were several reports on combination therapies, including those differentiating effects among specific subgroups.
Innovative prototype of a novel heart valve resection tool is based on foldable nickel-titanium (NiTi) blades.
Data show that Angiomax (bivalirudin) provides acute coronary syndrome patients who are undergoing PCI with similar results from ischaemic events and death versus standard therapy.
The FAME trial showed that routine measurements of fractional flow reserve during percutaneous coronary intervention compared to angiographic approach reduces rate of composite endpoint of death.
In JUPITER (Justification for the use of statins in prevention: an intervention trial evaluating rosuvastatin), a clinical trial enrolling individuals who do not currently qualify for statin therapy, rosuvastatin significantly reduced all-cause mortality by 20%.
The aim of this article is to review the incidence and all the possible causes and consequences of a suboptimal stent expansion.
Professor Alain Cribier is a pioneer in the field of aortic and mitral valvulopl...
Professor Fausto Pinto discusses his major influences, his career achievements and his passion for travelling, books and music.
The Editor of Vascular News, Professor Roger Greenhalgh, Imperial College, London, writes a personal appreciation of DeBakey.
0
A market report from BIBA Research shows that drug-eluting stents (DES) penetration rate in Western and Central Europe increased by 1.1% to 47.6% in the second quarter 2008.