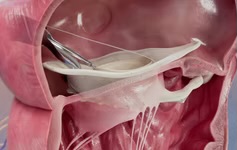
Edwards Lifesciences announced the global launch of the Carpentier-Edwards physio tricuspid annuloplasty ring for the treatment of tricuspid heart valve insufficiency. Edwards received both CE mark approval and FDA clearance.
“We designed the ring to offer surgeons confidence when treating tricuspid valve insufficiency, taking into account their feedback and a 40 year established body of scientific evidence on tricuspid valve reconstruction,” said Alain F Carpentier, professor and chairman emeritus of cardiovascular surgery at the Hôpital Europeen Georges Pompidou in Paris, France. “For example, the ring is designed to allow it to conform to the anatomy of the valve annulus and preserve its natural movement to facilitate adherence of the ring to the surrounding tissue, while restoring proper valve function.” Carpentier conducted the preliminary research for the ring.
The physio tricuspid annuloplasty ring features a three-dimensional waveform shape which allows the ring to conform to the natural shape of a patient’s tricuspid annulus during valve closure, and is designed with “Selective Flexibility” to allow the native valve to maintain its natural movement. The ring also incorporates a number of meaningful ease-of-implant features, including a distinct sewing ledge that facilitates intuitive suture placement and alignment.