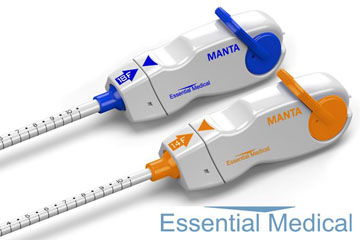
Teleflex has received premarket approval (PMA) from the US FDA for the Manta vascular closure device. A press release reports that the device is the first commercially available biomechanical vascular closure device designed specifically for large bore femoral arterial access site closure. Manta is indicated for closure of femoral arterial access sites while reducing time to haemostasis following the use of 10–20F devices or sheaths (12–25F outer diameter) in endovascular catheterisation procedures.
The SAFE MANTA IDE clinical trial, the largest US prospective multicentre, single-arm trial of a purpose-designed large bore femoral access site closure, demonstrated that the Manta device successfully achieves fast reliable biomechanical closure with rapid haemostasis, with all primary and secondary endpoints met. With its innovative design, the press release states, the Manta device has the potential to reduce bleeding complications and offset other procedural costs.
Zvonomir Krajcer (Texas Heart Institute, Houston, USA), the lead enroller and co-principal investigator of the SAFE MANTA IDE clinical trial, comments: “I am very encouraged by the results of the SAFE MANTA IDE clinical trial. The clinically proven major complication rate (as defined by the study protocol) of 5.3% and VARC-2 major vascular complications rate of 4.2% compare very favourably to suture mediated devices and the 24 second median time (65 second mean time) from deployment to haemostasis was impressive. We have been patiently waiting for this approval, are eager to use the MANTA Device commercially and look forward to the efficiencies it can provide.”
Greg Walters, co-inventor of the MANTA device and now vice president of access and closure in the Interventional business unit of Teleflex, states: “Our team has been working hard to obtain FDA premarket approval and were confident they would recognize the benefits that the MANTA™ Device can provide to the patient. We have had great success with the device in Europe over the last two years with over 10,000 units sold, and are thrilled to bring this innovative solution to patients in the USA and further fulfil this significant and previously unmet clinical need in the structural heart and endovascular space.”










