Edwards Lifesciences has received CE mark for the Mitris Resilia tissue valve replacement specifically designed for the mitral position.
Edwards Lifesciences has received CE mark for the Mitris Resilia tissue valve replacement specifically designed for the mitral position.
The valve is made with bovine pericardial tissue technology that is designed to reduce calcium build up on the valve. According to the company, this technology enables the valve to potentially last longer than conventional bioprosthetic valves.
Mitris Resilia has a saddle-shaped sewing cuff that mimics the asymmetric shape of the native mitral valve. The low-profile valve is based on the Edwards Perimount valve design with a nitinol wireform, which allows it to fold inward during implantation. The Mitris Resilia valve is visible under fluoroscopy, which facilitates potential future transcatheter interventions for patients.
Recent data on the Resilia tissue from the COMMENCE aortic trial reported encouraging results with low rates of structural valve deterioration (99.3% freedom from structural valve deterioration), clinically stable gradients and freedom from reoperation (97.2%) through seven years. Additionally, the COMMENCE mitral trial demonstrated clinically stable haemodynamics and freedom from structural valve deterioration (98,7%) at 5 years.
“Mitral valve disease is prevalent and patients experience the disease in various ways. For patients who need mitral valve replacement, the advanced Mitris Resilia valve, designed to mimic the native valve, is based on a trusted pericardial Perimount,” says Daniel Pereda Arnau (H Clínic de Barcelona, Barcelona, Spain). “As valve durability is becoming increasingly important, the integrity-preservation technology incorporated in the Resilia tissue that will potentially allow the valve to last longer, is critical.”
The Mitris Resilia valve received US Food and Drug Administration (FDA) approval in 2022 in the USA. The introduction of the Mitris Resilia valve into the portfolio demonstrates Edwards’ continuous investment in innovation for the benefits and the quality of life of patients.
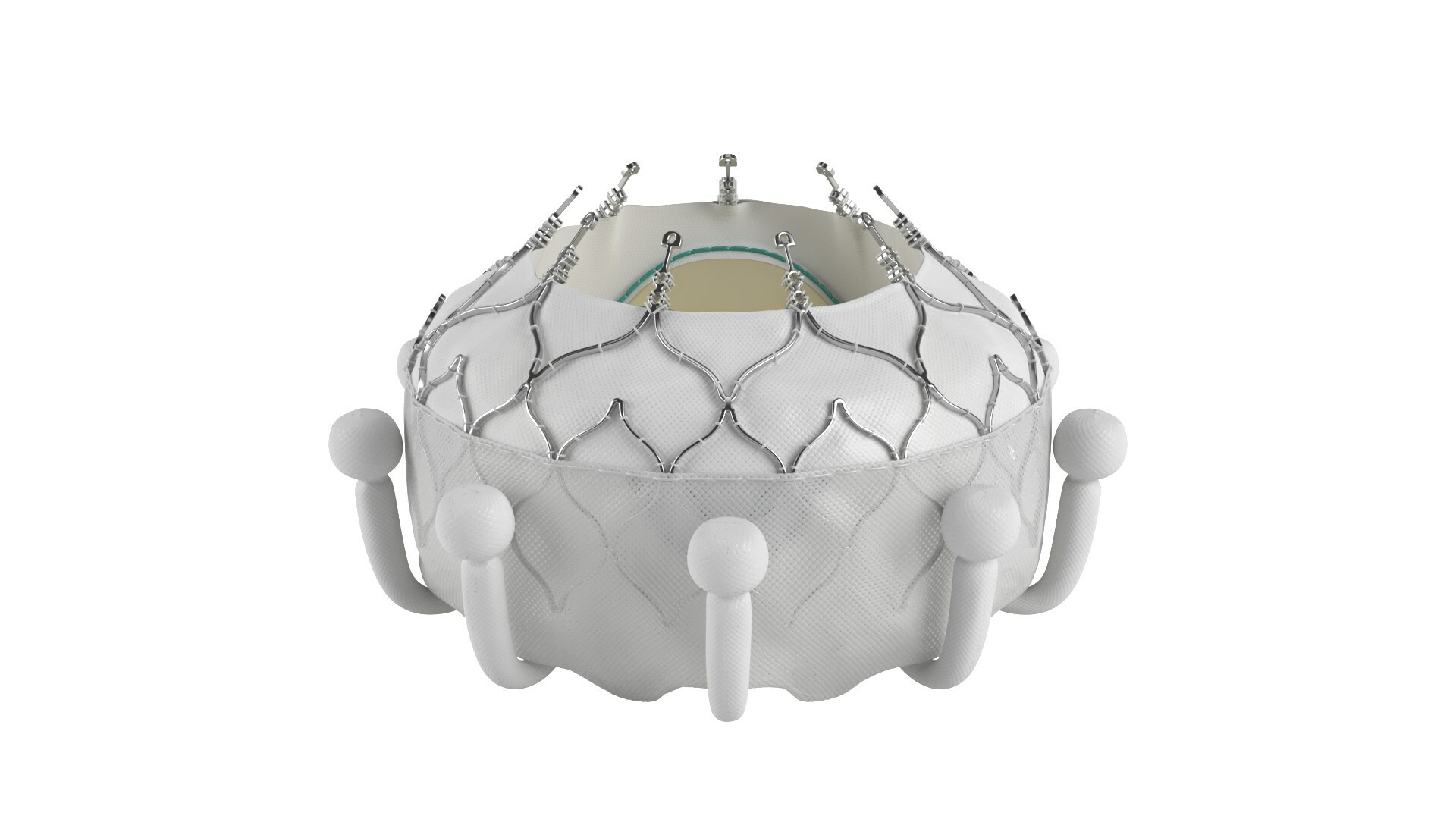
The company has also this month announced that the Evoque tricuspid valve replacement system received CE mark for the transcatheter treatment of eligible patients with tricuspid regurgitation (TR). The Evoque system is a transcatheter valve replacement therapy to receive regulatory approval to treat TR.
“Innovating for unmet patient needs is at the centre of everything we do at Edwards, which makes us especially proud to have received CE mark for this first-of-its-kind transcatheter tricuspid valve replacement therapy,” said Daveen Chopra, Edwards’ corporate vice president, transcatheter mitral and tricuspid therapies. “With the Evoque system’s approval, in addition to our current Pascal tricuspid system, we are now able to provide a broader array of much-needed treatment options for appropriate tricuspid disease patients in Europe.”
The Evoque system is comprised of a nitinol self-expanding frame, intra-annular sealing skirt, and tissue leaflets made from the same bovine pericardial tissue as the company’s current heart valves. The EVOQUE valve will be available in three sizes, all delivered through a low-profile transfemoral 28F system.
“The Evoque system is able to fully replace the tricuspid valve, virtually eliminating tricuspid regurgitation in a wide range of anatomies,” said Philipp Lurz (University of Mainz, Mainz, Germany) and European principal investigator for the TRISCEND II study. “The significant improvements in patients’ quality-of-life are remarkable, now offering a therapy to many patients who previously had no treatment options.”