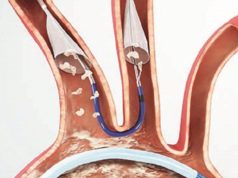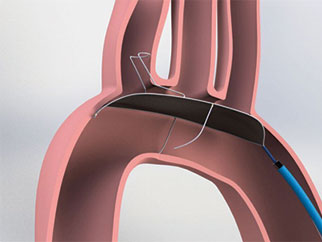
Keystone Heart announced the launch of phase II of the REFLECT trial, which is evaluating the next-generation TriGuard 3 cerebral protection device. According to a press release, the device is being assessed for its ability to protect the brain from emboli during transcatheter aortic valve implantation (TAVI) and, thus, minimise the risk of stroke and cerebral damage.
The REFLECT trial is a multicentre, prospective, randomised, clinical study designed to assess the safety and efficacy of comprehensive cerebral protection from emboli released during cardiovascular procedures. Phase I of this trial, which remains blinded, enrolled 258 patients and used the TriGuard HDH—an earlier generation device that has the CE mark—and phase II of the trial is now initiating enrolment of up to 275 additional patients. The trial is designed to definitively address the role of comprehensive cerebral embolic protection in improving the safety of TAVI. Trial completion is targeted for October of this year with FDA submission shortly thereafter.
Tamim Nazif (Columbia University Medical Center, New York, USA), comments: “Knowing what we now know about the significant risks of stroke and other neurological injury associated with TAVI and other cardiovascular procedures, there is a clear unmet clinical need for cerebral embolic protection devices that provide complete protection of all three cerebral branches for patients undergoing these procedures. In our initial experience with TriGuard 3, the device appears to offer reliable, complete cerebral embolic protection, while also offering substantial improvements in ease of use and generalisability. We look forward to completing the REFLECT trial and advancing our understanding of the benefits of this device in protecting our patients at risk of cerebral damage during heart procedures.”
The press release reports that pooled analysis from the Keystone Heart DEFLECT clinical studies demonstrated that brain protection with TriGuard HDH was able to significantly reduce the rate of VARC 2 defined strokes at 30 days (0% vs 6%), the worsening of neurological findings NIHSS (0% vs 19%), and showed higher incidence of totally clean brains (28% vs 8%).
Chris Richardson, president and CEO, Keystone Heart, says: “We believe that it is imperative to bring a cerebral embolic protection device to the market that: protects all areas of the brain while minimizing device interaction; avoids any manipulation of the arch vessels or an additional access site; is anatomy independent for universal patient application; and comes at a price point that will allow broad adoption for the majority of TAVI patients. TriGuard 3 is designed to accomplish just that.”