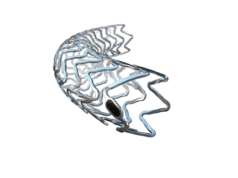
The US FDA have approved Biotronik’s drug-eluting stent Orsiro. A press release states that Orsiro is the first and only ultrathin drug-eluting stent to outperform the clinical standard (Xience, Abbott). The device received CE marking in 2011 and has been used to treat more than one million patients worldwide to date.
According to the press release, the BIOFLOW-V trial demonstrated significantly lower rates for target lesion failure and target vessel myocardial infarction at 12 months with Orsiro compared with Xience in a large, complex patient population (n=1,344). At two years, treatment with Orsiro further improved event rates in comparison to Xience, including a significant reduction in target lesion revascularisation and spontaneous myocardial infarction. The two-year results—presented at 2018 Transcatheter Cardiovascular Therapeutics (TCT) meeting (21–25 September, San Diego, USA) and published in the Journal of the American College of Cardiology—showed a 37% lower target vessel failure rate in favour of Orsiro (7.5% vs. 11.9% target lesion failure rate; p=0.015), a 47% lower ischaemia-driven target lesion revascularisation rate, and a 70% lower rate of spontaneous myocardial infarction (landmark analysis 31 days to two years follow-up).
David Kandzari (Piedmont Heart Institute, Atlanta, USA), BIOFLOW-V US principal investigator, says: “Orsiro has set a new standard for safety and efficacy clinical endpoints, including statistically lower target lesion revascularisation and target vessel myocardial infarction rates. BIOFLOW-V data are the best clinical outcomes witnessed with a modern drug-eluting stent. It was largely thought that efficacy findings were unsurpassable, but Orsiro proves we can further reduce event rates with meaningful innovation,”
For use in percutaneous coronary intervention (PCI) procedures, the cobalt chromium metal stent elutes sirolimus via Biolute—Biotronik’s biodegradable polymer coating. Beneath the biodegradable layer is proBio, a passive coating on the bare metal surface, designed to reduce nickel ion release. The Orsiro stent system provides ultrathin stent struts without compromising radial strength, and a low crossing profile for easier lesion cross in complex PCI. It is available in 52 sizes ranging from 2.25mm to 4mm in diameter and lengths up to 40mm, the longest available in the USA.
Ryan Walters, president at Biotronik, states: “The FDA approval of Orsiro changes the dynamic of what had become a highly commoditised drug-eluting stent market. We designed Orsiro for use even in challenging cases with features that make it unlike any other drug-eluting stent in the world. Hospital administrators now have available a drug-eluting stent that shows improved clinical event rates and interventionalists can rely on Orsiro’s deliverability to treat complex lesions and challenging subgroups to achieve unprecedented patient outcomes. Patients, physicians and health systems deserve the best, and that is exactly what we are bringing to the US market.”