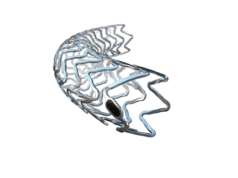
Data presented during EuroPCR (22 May – 25 May, Paris, France) provide further evidence on its continued safety and efficacy, also in a more complex population. Stefan Verheye (Antwerp Cardiovascular Institute, Antwerp, Belgium) presented results on the first 200 patients from the cohort of 1,065 patients included in the BIOSOLVE-IV registry.
The data show comparable target lesion failure rates (4.6%) to second-generation drug-eluting stents at 12 months and only one case of scaffold thrombosis (after DAPT interruption before the event). These promising outcomes included 14.5% of NSTEMI patients, a challenging patient group. Michael Kang-Yin Lee (Queen Elizabeth Hospital, Hong Kong, China), principal Investigator of the BIOSOLVE-IV study, comments: “The new data give insights in a more complex population and add to the body of evidence supporting the efficacy of Magmaris.”
Additionally, new optical coherence tomography data at three years from BIOSOLVE-II was presented by Michael Joner (German Heart Center, Munich, Germany. The presented data confirmed earlier preclinical findings on Magmaris showing reduced signs of neoatherosclerosis.
Alexander Uhl, senior vice president corporate marketing at Biotronik, says: “As more evidence of the benefits of magnesium as a scaffold material emerges, supported by the results of our ongoing clinical programme, Biontronik will further invest in bioresorbable magnesium technology. This investment underlines our belief that the resorbable magnesium scaffold has a significant role to play in interventional cardiology. We firmly expect that evidence will support the relevance of the scaffold in wider indications and patient populations.”