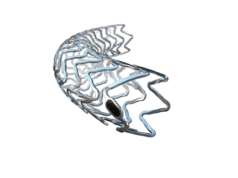
 Biotronik has announced the start of enrolment in a coronary stent trial aiming at assessing the safety and clinical performance of a new coronary stent (Biovtiesse) in de novo coronary artery lesions.
Biotronik has announced the start of enrolment in a coronary stent trial aiming at assessing the safety and clinical performance of a new coronary stent (Biovtiesse) in de novo coronary artery lesions.
On September 28, Marco Moccetti (Cardiocentro Ticino, Lugano, Switzerland) and subsequently (on the same day) Lorenz Raeber (University Hospital Bern, Switzerland) successfully implanted the new polymer-free coronary stent in their first patients.
According to a press release, the novel device combines the proven PRO-Kinetic Energy stent features with its ultrathin struts, with a new anti-proliferative limus drug designed to prevent excessive cell growth.
Trial details
The BIOVITESSE prospective, multicentre, first-in-man trial will investigate the treatment of coronary de novo lesions. Primary endpoints are strut coverage at one month and in-stent late lumen loss at nine months. Strut coverage will be assessed by optical coherence tomography (OCT) analysis. Late lumen loss will be assessed by quantitative coronary angiography (QCA) assessment.
Daniel W Buehler, Biotronik president, Vascular Intervention, comments: “As an established and trusted provider of a leading vascular intervention portfolio, Biotronik has once again demonstrated its commitment to innovation by the development of this new coronary stent system. Our Biovtiesse stent system’s ease of use reflects our long-term expertise in developing coronary stents, which allows us to continue to improve performance and provide enhanced treatment options.”
The BIOVITESSE study will run in five centres in Switzerland until August 2019.