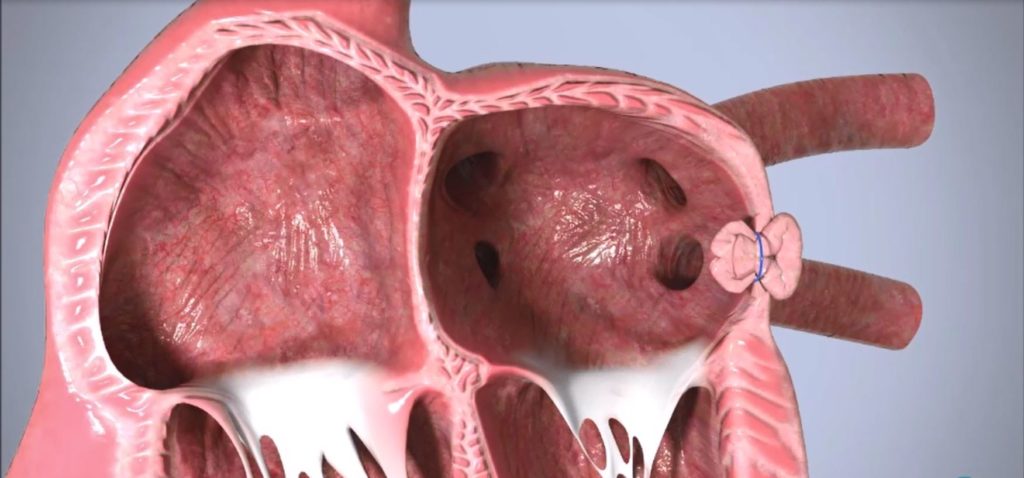
Append Medical, developer of the Appligator implant-free left atrial appendage (LAA) occlusion device, today announced completion of its first pre-clinical chronic procedures. The procedures were completed with the research organisation IMMR and pave the way for the first-in-human trial of the device, Append Medical said in a press release.
“These first procedures provide an important validation for our approach, which is intended to significantly simplify the goal of sealing the LAA and preventing stroke in atrial fibrillation (AF) patients without leaving any implant in the heart,” said Zachi Berger, founder and CEO of Append Medical. “Organising this trial during the current pandemic was not a simple task and we would like to thank the relevant French bodies as well as the IMMR team for allowing us to complete this trial while complying with COVID-19 restrictions.”
“My team and I found the procedure to be elegant and straightforward toward continuing to make LAA exclusion more accessible,” said Nicolas Borenstein, scientific and technical director at IMMR. Both animals are doing fine so far, and the post-procedure images are spectacular. Congratulations to the Append team on this achievement.”
The Append Medical Appligator is designed to reduce stroke risk in AF patients by excluding the LAA to prevent blood clot leakage with a minimally invasive transseptal intervention leaving no implant behind. The Appligator device utilises natural tissue manipulation to achieve complete LAA closure through invagination of the LAA into the left atrium. This approach is designed to minimise device-related thromboembolism risk by leaving only a suture—and no metal implant—at the closure site.
Append Medical operates as part of MEDX Xelerator in Israel. MEDX Xelerator’s partners include MEDX Ventures, Boston Scientific., Intellectual Ventures and Sheba Medical Center.










