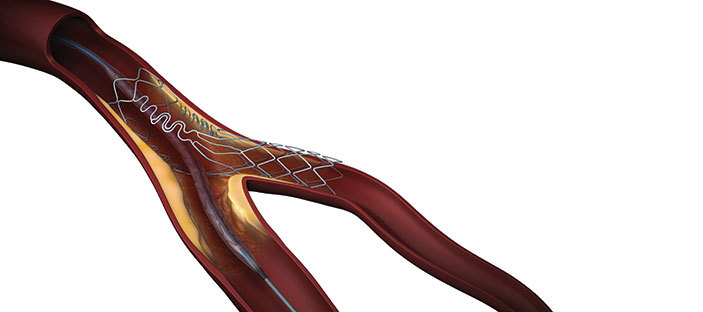
The US Food and Drug Administration (FDA) has approved Tryton Medical’s Premarket Approval (PMA) application for the Tryton side-branch stent for the treatment of coronary bifurcation lesions involving large-side branches (appropriate for a ≥2.5mm stent). The Tryton side-branch stent is the first dedicated bifurcation device to receive regulatory approval in the USA.
In a post hoc analysis of a randomised investigational device exemption (IDE) clinical trial, treatment with the Tryton side-branch stent in the intended population of patients with large side branches (appropriate for a ≥2.5mm stent) reduced the need for additional bailout stenting (0.7% vs. 5.6%, p=0.02) and led to statistically significant lower side branch per cent diameter stenosis at nine month follow up (30.4% vs. 40.6%, p=0.004) when compared to provisional stenting. The post hoc analysis also showed comparable major adverse cardiovascular events (MACE) and myocardial infarction (MI) rates versus provisional stenting at three years.
The safety profile of the Tryton side-branch stent was validated in a confirmatory study that compared patients treated with the Tryton stent to a performance goal based on performance of the control arm from the randomized IDE clinical trial. The confirmatory study met its pre-specified primary endpoint, periprocedural myocardial infarction (PPMI), which was within its non-inferiority margin (primary endpoint: 10.5% + 95% CI vs. 17.9%, p=0.01).
“Treatment of complex lesions at the site of a bifurcation has historically been inconsistent, with results varying depending on the procedure and the experience of the interventionist,” says Aaron Kaplan, professor of Medicine at Dartmouth Hitchcock Medical Center, Lebanon, USA, and chief medical officer of Tryton Medical. “A predictable bifurcation solution helps alleviate some of the stress in these procedures by limiting variability and reducing the need for bailout stenting.”
Tryton has also signed a strategic distribution agreement with Cardinal Health enabling Cordis, its interventional vascular business, to be the exclusive distributor of the Tryton side branch stent in the USA.
The Tryton side-branch stent is available in multiple device diameters (2.5mm to 3.5mm in the side branch) and is compatible with any conventional drug-eluting stent in the main vessel.