 One-year follow-up results of the randomised SOLVE-TAVI trial showed similar rates of all-cause death, cardiovascular death, moderate or severe prosthetic valve regurgitation, and permanent pacemaker implantation in balloon expandable valves and self-expandable valve treated patients, but found that stroke occurred more frequently the balloon expandable valve group. The findings were presented by Hans-Josef Feistritzer (University Hospital, Leipzig, Germany) during a late breaking clinical science session at TCT Connect 2020 (14–18 October).
One-year follow-up results of the randomised SOLVE-TAVI trial showed similar rates of all-cause death, cardiovascular death, moderate or severe prosthetic valve regurgitation, and permanent pacemaker implantation in balloon expandable valves and self-expandable valve treated patients, but found that stroke occurred more frequently the balloon expandable valve group. The findings were presented by Hans-Josef Feistritzer (University Hospital, Leipzig, Germany) during a late breaking clinical science session at TCT Connect 2020 (14–18 October).
Opening his presentation Feistritzer said that during the last decade transcatheter aortic valve implantation (TAVI) has developed as the standard strategy in patients with severe symptomatic aortic stenosis at high to intermediate risk. “Major innovations in TAVI design led to relevant technical and clinical improvements, for instance lower pacemaker rates, improved paravalvular regurgitation and also vascular complications,” he said. “Nevertheless there is limited data derived from randomised trials regarding direct valve comparisons.“
He added that regarding anaesthesia strategy, it is important to note that around 50% of TAVI procedures are performed under local anaesthesia with conscious sedation, and registry data has shown that this strategy is associated with lower morbidity and mortality, shorter ICU and overall hospital stays and shorter procedure times.
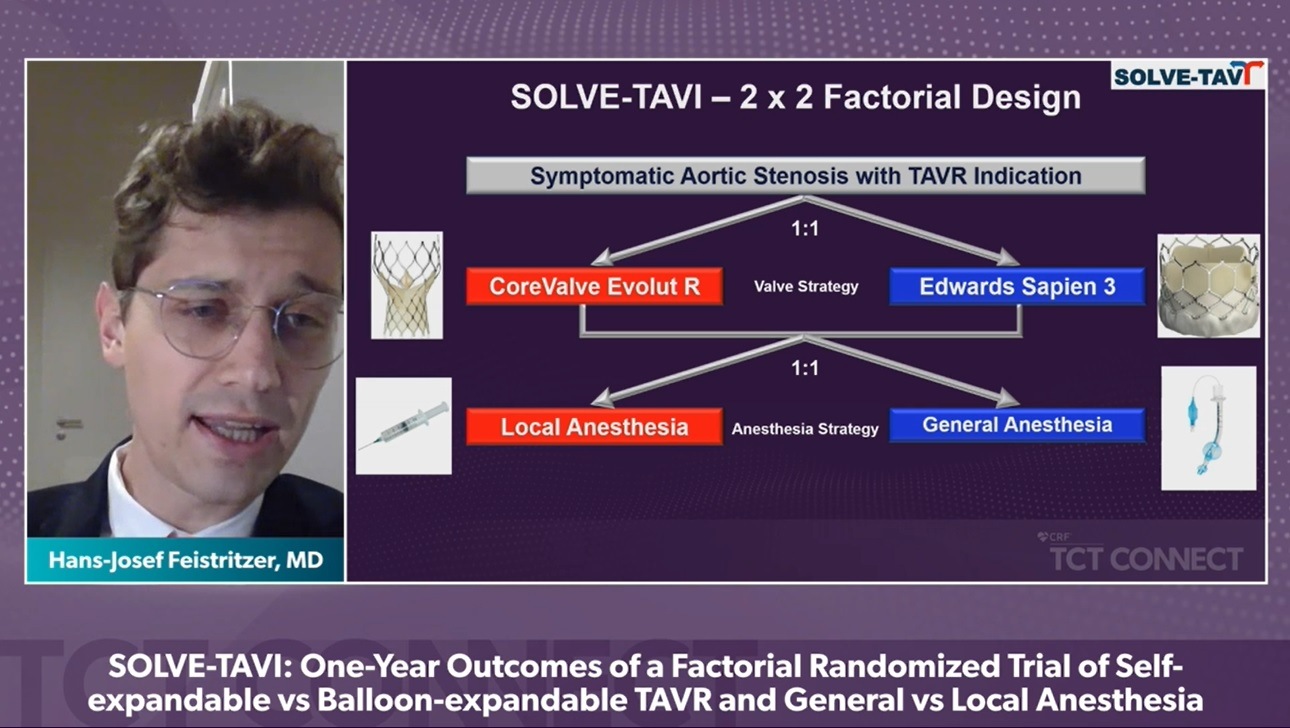
In the randomised, multicentre, 2×2 factorial, open-label SOLVE-TAVI trial 447 patients with severe, symptomatic aortic stenosis were randomly assigned to transfemoral TAVI using either the Sapien 3 (Edwards Lifesciences) balloon-expandable device, or the CoreValve Evolut R (Medtronic) self-expandable valve, as well as being randomised to either general or local anaesthesia. Patients were followed-up for 1 year for clinical outcome assessment and evaluation of echocardiographic valve function. The trial was powered to show equivalence of the primary endpoint for the valve comparison strategy as well as for the anaesthesia comparison, Feistritzer explained.
The primary endpoint of the valve comparison strategy was the combined endpoint of all-cause mortality, stroke, moderate or severe prosthetic valve regurgitation and permanent pacemaker implantation at 30 days. For the anaesthesia strategy, the primary endpoint at 30 days was also a composite endpoint, it included all-cause mortality, stroke, myocardial infarction, infections requiring antibiotic treatment, and acute kidney injury.
The results presented by Feistritzer showed that at one-year follow-up, rates of all-cause death (17.0% vs. 17.6%; hazard ratio [HR] 0.96, 95% confidence interval [CI] 0.60 to 1.55), cardiovascular death (1.8% vs. 0.5%; HR 3.89, 95% CI 0.44 to 34.67), moderate or severe prosthetic valve regurgitation (4.5% vs. 7.0%; HR 0.63, 95% CI 0.27 to 1.45), and permanent pacemaker implantation (20.2% vs. 24.7%; HR 0.79, 95% CI 0.53 to 1.16) were similar between the balloon expandable and self-expandable valve groups, but the stroke rate was higher in the balloon expandable valve treated patients (6.9% vs. 1.0%; HR 7.13, 95% CI 1.62 to 31.32).
Feistritzer said: “We showed in the SOLVE TAVI trial that in patients with severe symptomatic aortic stenosis undergoing transfemoral TAVI, newer generation self-expanding compared to balloon expandable valves showed similar rates of a composite endpoint of all-cause death, stroke, paravalvular leakage and permanent pacemaker implantations. However, the stroke rates were higher in the balloon expandable valves.”
Regarding the anaesthesia strategy Feistritzer noted that the results showed that local anaesthesia with conscious sedation compared to general anaesthesia resulted in a similar rate of all-case death, cardiovascular death, stroke, infarctions, infections and acute kidney injury.













