Tag: CardioCel
Admedus sells CardioCel and VacsuCel to focus on TAVI
Admedus has sold the distribution rights to its CardioCel and VascuCel product range for $A36.2 ...
ECHSA 2019: Shape matters and is an innovation
Martin Kostelka (Leipzig, Germany), who was the first to use the CardioCel 3D (Admedus) product ...
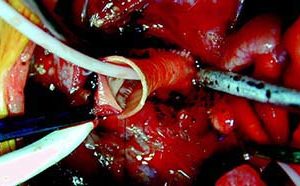
CardioCel benefits patients with different anatomies for intracardiac application
Speaking to Cardiovascular News at the ECHSA 2019 meeting (European Congenital Heart Surgeons As...
CardioCel 500—Long term experience with no calcification
Giovanni Stellin (Padova, Italy) talks to Cardiovascular News at the ECHSA 2019 meeting (Europea...
CardioCel use improves management of critically ill neonates
Tomasz Mroczek (Kraków, Poland) talks to Cardiovascular News at ECHSA 2019 (European Congenital...
ECHSA 2019: CardioCel acellular bioscaffold handles well
Caner Salih (London, UK) talks to Cardiovascular News about the CardioCel bioscaffold (Admedus) ...
Admedus’ announces favourable five-year outcomes for its CardioCel bioscaffold
Admedus has announced positive data from an independent study into the performance of its tissue...
CardioCel 3D and VascuCel receive regulatory approval in Europe
Admedus has received the CE mark for its CardioCel 3D product portfolio and its next-generation ...
CardioCel now approved for use in the UAE
Admedus has announced that, in collaboration with its regional partner Genpharm, it has received...
Long-term study indicates no calcification with CardioCel after eight years
Long-term data from a Phase II extension study for the repair of congenital heart defects in pae...