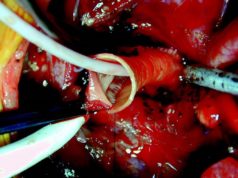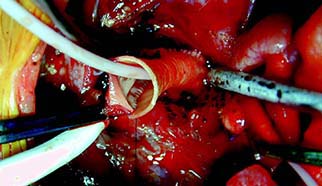
Admedus has received the CE mark for its CardioCel 3D product portfolio and its next-generation collagen bioscaffold VascuCel in Europe. According to a press release, this regulatory approval is an important step forward as the company continues to increase global sales of its adapt tissue technology and to create future value through the rollout of its novel 3D technology.
The press release reports that the CardioCel 3D product portfolio is the world’s first and only shaped and proven fully acellular collagen bioscaffold. It is designed for highly complex congenital defect repairs and enables cardiac surgeons to achieve a more natural shape during aortic arch reconstruction; combined with the unique clinical benefits of calcification resistance, which is one of the most significant problems that clinicians face post procedure.
Admedus intends to commence the first phase of its commercial launch of CardioCel 3D later this month, initially working with selected European Key Opinion Leaders already familiar with the clinical benefits of Adapt tissue technology. In the subsequent phase, there will be a broader peer to peer education programme to benefit more patients.
Tomasz Mroczek (Department of Paediatric Cardiac Surgery, University Children’s Hospital, Jagiellonian University Medical College, Kraków, Poland says: “After our initial positive experience with Adapt, we are excited to have the choice of these 3D shaped products that will produce better reconstructions for complex aortic arch repair surgeries.”
Wayne Paterson, CEO of Admedus, says: “Obtaining CE mark approval for the 3D portfolio is a further endorsement of the medical need and effectiveness of our 3D shaped collagen bioscaffold; a world first technology with proven anticalcification properties and zero DNA. The approval of the 3D portfolio in Europe further reinforces the company’s focus on global strategic growth with geographic expansion into key markets and portfolio expansion of ADAPT® tissue technology products.”