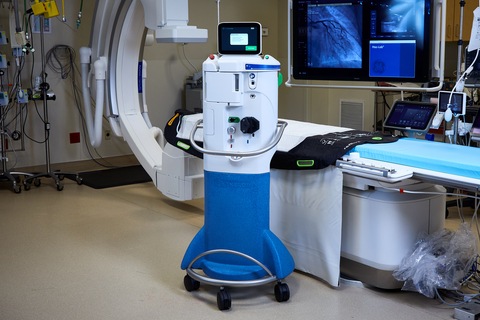
 Zoll Medical has announced that the first patient has been enrolled in the AMIHOT III post-approval study evaluating TherOx SuperSaturated Oxygen (SSO2) therapy for the treatment of left anterior descending (LAD) ST-Elevation Myocardial Infarction (STEMI).
Zoll Medical has announced that the first patient has been enrolled in the AMIHOT III post-approval study evaluating TherOx SuperSaturated Oxygen (SSO2) therapy for the treatment of left anterior descending (LAD) ST-Elevation Myocardial Infarction (STEMI).
The patient, treated at WakeMed Health & Hospitals in Raleigh, USA, received SSO2 Therapy, the first US Food and Drug Administration (FDA)-approved therapy to significantly reduce the size of infarct following percutaneous coronary intervention (PCI).
TherOx SSO2 Therapy is currently indicated for patients who suffer LAD STEMI treated within six hours of symptom onset. SSO2 therapy has been shown to reduce relative infarct size by 26% over standard of care, Zoll Medical said in a press relase. One-year follow-up data on patients treated with SSO2 therapy showed reductions in heart failure hospitalization and mortality.
AMIHOT III is a randomised, post-approval evaluation that will validate the benefits of SSO2 therapy over current standard of care alone and provide insight into the mechanisms that lead to improved outcomes for LAD STEMI patients. Additional endpoints include incidence of microvascular obstruction (MVO), and outcomes measures such as heart failure readmissions and quality-of-life measures. The study will randomise 434 patients across sites in the USA and Europe. The study’s principal investigator is James Blankenship (University of New Mexico, Albuquerque, USA).
“WakeMed strives to be on the cutting edge of cardiovascular care, and I am excited that our team has enrolled the first AMIHOT III patient in this study to further confirm SSO2 Therapy’s benefits over standard of care in the treatment of severe heart attacks,” said Frances Wood (WakeMed Health & Hospitals, Raleigh, USA). “Based on our experience with the IC-HOT trial that helped SSO2 achieve FDA approval, we believe this therapy will have a strong impact in improving outcomes and reducing mortality for heart attack patients.”