Royal Philips has announced the official start of the DEFINE GPS study, with the first patient being enrolled by the study’s principal investigator, Allen Jeremias at St. Francis Hospital, New York, USA.
The global, multicentre, prospective, randomised controlled DEFINE GPS study will investigate if guidance by instantaneous wave-free ratio (iFR) measurements co-registered on the angiogram demonstrates superior outcomes and improves the cost-effectiveness of percutaneous coronary intervention (PCI) procedures. DEFINE GPS employs an adaptive study design, estimated to include up to 3,200 participants across 100 sites worldwide and will be one of the largest studies ever sponsored by Philips.
European and US clinical guidelines already endorse the use of physiological measurement of coronary function in PCI procedures, with iFR and fractional flow reserve (FFR) measurements being used to diagnose the significance of a narrowed coronary artery and determine patient selection for treatment. While iFR uses the same pressure guide wires and equipment as FFR, it avoids the use of hyperaemic agents such as adenosine that can adversely affect some patients.
The DEFINE GPS (Distal Evaluation of Functional performance with Intravascular sensors to assess the Narrowing Effect: Guided Physiologic Stenting) study will evaluate the use of iFR measurements in combination with Philips Image Guided Co-Registration System—SyncVision—to enhance PCI guidance and provide superior treatment outcomes. DEFINE GPS is a follow-up to the DEFINE PCI study—a one-year trial sponsored by Philips that evaluated the potential of treating residual ischaemia in order to improve clinical outcomes for coronary stent patients.
“The DEFINE GPS trial, designed to explore the promising hypotheses that emerged from DEFINE PCI, is a landmark trial that heralds the promise of post-PCI physiologic assessment,” said Gregg W Stone, chairman of the DEFINE GPS trial and the director of academic affairs for the Mount Sinai Heart Health System, New York, USA. “While the benefits of physiology prior to PCI are unquestioned, this large-scale trial will definitively demonstrate whether after an angiographically successful PCI procedure the identification and treatment of unsuspected residual ischaemia by routine iFR assessment can further improve patient event-free survival.”
“Conducting rigorous clinical science is how we advance patient care, and, like the seminal FAME study that was carried out over 10 years ago, I believe DEFINE GPS has the potential to change the current standard of care in PCI,” said Jeremias. “PCI has made a major positive impact on many coronary artery disease patients’ lives, but when we look back at all the major, high-quality stent trials over the past 20 years we see that around 20‒30% of patients continue to have recurring chest pain at one year after receiving treatment. With DEFINE GPS we will be able to definitively determine if a physiology-based PCI approach results in superior patient outcomes compared to standard angioplasty.”
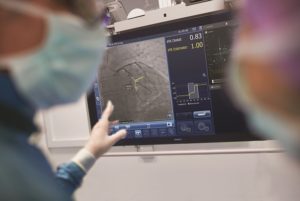
PCI is an image-guided, minimally-invasive treatment to open a coronary artery blockage that is causing ischaemia. Under the current standard of care, clinicians navigate a balloon catheter and coronary stent to the treatment area using interventional X-ray guidance. In the DEFINE GPS study, an iFR pullback measurement, which uses pressure wires to map the physiological profile of disease distribution along the length of the affected vessel, will be overlaid on the angiogram to provide more precise information on where to treat within the vessel. The study will also use iFR to measure if the treatment succeeds in restoring sufficient blood flow to prevent ischaemia or if further treatment is warranted.
“iFR continues to be adopted in clinical practice, with mounting evidence that this innovative technology contributes to improved outcomes, reduced costs and enhanced patient experiences,” said Chris Landon, senior vice president and general manager image guided therapy devices at Philips. “This major study will provide a definitive answer to the question of whether a functional guidance strategy throughout the procedure demonstrates superior outcomes and reduces costs. The study has the potential to drive a significant improvement in clinical practice, and it is a prime example of Philips’ commitment to providing a strong evidence base for the benefits of its healthcare innovations.”
The DEFINE GPS study is sponsored by Philips, with the Cardiovascular Research Foundation overseeing core lab and clinical event committee activities.