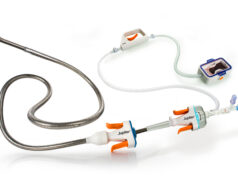
Boston Scientific Corporation has received CE mark approval for the Rebel platinum chromium coronary stent system, the company’s latest generation bare metal stent for the treatment of coronary artery disease.
According to a company release, the Rebel stent system was developed to expand the Boston Scientific platinum chromium customised architecture family of stents, and offers physicians the same stent platform as the Promus Premier drug-eluting stent but without the everolimus drug. Bare metal stents continue to play an important role in the treatment of coronary artery disease and represent a significant portion of the global stent market.
“I am glad to have a bare metal stent that performs like the Premier drug-eluting stent but allows me to treat patients who are not candidates for drug-eluting stent therapy,” says Didier Carrie, an investigator for the OMEGA clinical trial at Centre Hopital Universitaire, Rangueil, France. “In addition to its great visibility and deliverability, the platinum chromium stent architecture features low recoil that is particularly important for patients treated with bare metal stents.”
The press release adds, the Rebel stent system features unparalleled visibility, low recoil, exceptional radial strength and fracture resistance, while improving axial strength and deliverability. Its enhanced low-profile delivery system also features a shorter, more visible tip, a dual-layer balloon and a bi-segment inner lumen catheter designed to facilitate precise stent delivery across challenging lesions.
Data from the OMEGA clinical trial evaluating the platinum chromium bare metal stent system were presented in February by John C Wang, Medstar Union Memorial Hospital, Baltimore, USA, at the Cardiovascular Research Technologies conference in Washington, DC. OMEGA is a single arm, multicentre trial in the US and Europe. OMEGA trial data are expected to support a US Food and Drug Administration regulatory submission.
In the US, the Rebel stent system is an investigational device and not available for sale.