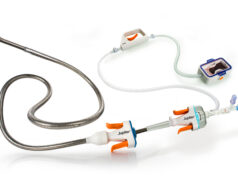
CardiacAssist has launched the Protek17 arterial cannula, which it says is a “key component” of its TandemHeart temporary circulatory support platform. It adds that the new cannula has been designed for improved patient safety and ease of use, with new features including suture wings for secure attachment to the patient and a rubber stop to prevent over-insertion.
According to a press release, the Protek17 cannula is FDA-cleared and has been successfully deployed in multiple TandemHeart cases during a limited commercial release in 2013. The device is now available for full commercial distribution.
“The Protek17 cannula represents a major milestone for our company and is the first in a family of products we plan to launch in the near future,” said John Marous, president and chief executive officer of CardiacAssist. “We will continue to focus on new product development and the advancement of clinical science to improve the quality of circulatory support therapy across a wide spectrum of patient needs.”