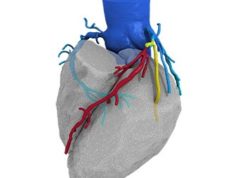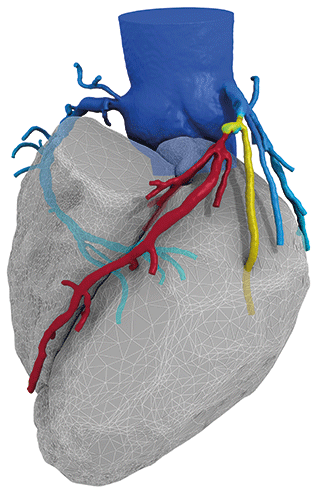
HeartFlow has announced that the Health Care Service Corporation (HCSC)—that operates Blue Cross and Blue Shield plans in Illinois, Montana, New Mexico, Oklahoma and Texas (USA)—has issued a medical policy for the HeartFlow FFRct analysis system.
Under this policy, the HCSC has determined that the non-invasive technology, which is designed to help clinicians diagnose and treat patients with suspected coronary artery disease, may be considered medically necessary to guide decisions about the use of invasive coronary angiography in patients with stable chest pain at intermediate risk of coronary artery disease.
Additionally, a press release reports, BlueCross BlueShield companies in Tennessee, Louisiana and South Carolina recently issued a positive medical policy for the HeartFlow FFRct analysis. After reviewing the technical performance, diagnostic accuracy, clinical utility and clinical benefits, all three entities concluded that the use of the HeartFlow FFRct analysis following a coronary computed tomography (CT) angiogram may be considered medically necessary for its members with stable chest pain and results in meaningful improvements in the net health outcome.
John H Stevens, president and chief executive officer of HeartFlow, comments: “These new positive medical coverage decisions follow close on the heels of the Centers for Medicare & Medicaid Services’ decision to begin reimbursing for the HeartFlow FFRct Analysis under a New Technology Ambulatory Payment Classification effective January 1, 2018. With numerous other private payers already having issued coverage decisions, this news means that approximately 90 million Americans covered by commercial health plans and 55 million Medicare beneficiaries will have access to our pioneering technology.”