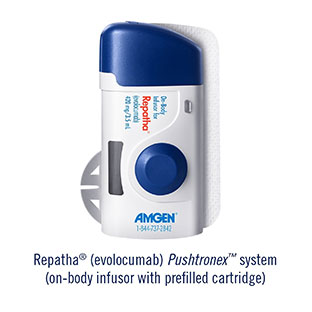
Amgen has received FDA approval for its Repatha (evolocumab) Pushtronex system (on-body infusor with prefilled cartridge)—a new, monthly single-dose administration option. The Pushtronex system is a hands-free device designed to provide 420mg of Repatha in a single dose, making Repatha the first and only PCSK9 inhibitor to offer a monthly single-dose delivery option.
A press release reports that in the USA, Repatha is indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease, who require additional lowering of low density lipoprotein (LDL) cholesterol. It can also be used as an adjunct to diet and other LDL-lowering therapies for the treatment of patients with homozygous familial hypercholesterolemia over the age of 13, who require additional lowering of LDL cholesterol. The effect of Repatha on cardiovascular morbidity and mortality has not been determined.
According to the press release, the new, single-use device was developed in collaboration with West Pharmaceutical Services, based on the SmartDose technology platform, to provide patients with an additional dosing option for Repatha treatment. The device adheres to the body and patients are hands free during administration. Patients are able to perform moderate physical activities (such as walking, reaching or bending) as the 420mg of Repatha is delivered subcutaneously.
Sean E Harper, executive vice president of Research and Development at Amgen, says: “The Pushtronex system exemplifies Amgen’s continued innovation and commitment to patients. Repatha is the only PCSK9 inhibitor with an approved monthly dose, and now the only one with a monthly single-dose administration. The FDA approval of the Pushtronex system offers another delivery option to patients who need the additional LDL cholesterol lowering that Repatha can provide.”












