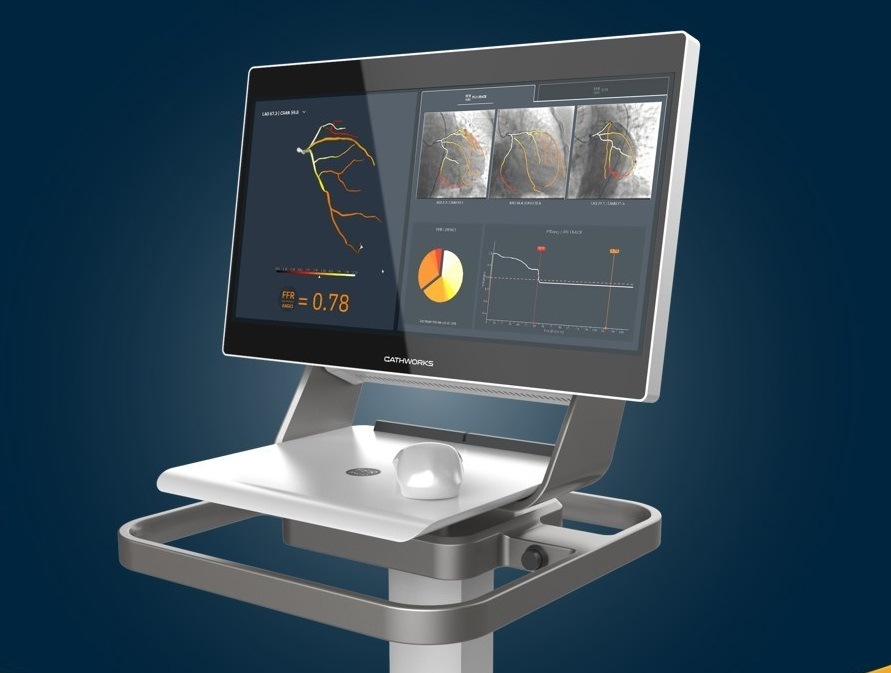
CathWorks has announced approval of the fourth generation CathWorks FFRangio system by the Japanese Pharmaceuticals and Medical Devices Agency (PMDA). The FFRangio system is also commercially available in the USA and Europe.
“The FFRangio System enables us to quickly perform a comprehensive physiologic assessment of coronary artery disease without the need for invasive pressure wires or hyperaemic agents, providing significant benefits to clinicians and patients,” said Yutaka Hikichi (Saga-Ken Medical Center Koseikan, Saga, Japan). “The approval of the fourth-generation application offers significant automation and enhancements, further simplifying the utilisation of the platform.”
“Coronary artery disease is a life-threating disease, and it is critical that we can quickly, effectively, and cost-effectively diagnose the disease and determine the appropriate treatment plan,” said Mamoru Nanasato (Sakakibara Heart Institute, Tokyo, Japan). “We have been able to experience first-hand how FFRangio can transform the diagnosis and treatment of our patients with coronary artery disease.”
The CathWorks FFRangio system has been the first non-invasive device of its kind to receive Japan PMDA and Ministry of Health, Labour and Welfare (MHLW) approval for diagnosis of functional ischemia for patients with ischemic coronary artery disease, CathWorks said in a press release. The fourth-generation application includes automation and enhancements, while offering the same diagnostic accuracy when compared to invasive wire-derived FFR, the press release adds.











