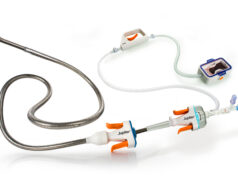
Biosensors has announced the enrolment of the first patient in the RISE SC study, a single arm, prospective, multicentre first-in-human trial to evaluate the acute safety and device success of the Rise SC coronary balloon when used as a dilatation device in the stenotic portion of a coronary artery or bypass graft in patients with coronary artery disease (CAD).
Biosensors has announced the enrolment of the first patient in the RISE SC study, a single arm, prospective, multicentre first-in-human trial to evaluate the acute safety and device success of the Rise SC coronary balloon when used as a dilatation device in the stenotic portion of a coronary artery or bypass graft in patients with coronary artery disease (CAD).
The trial is targeting CE-mark approval for the Rise semi-compliant balloon (SCB) and will include three investigational centres in Switzerland led by Marco Roffi, director of the Interventional Cardiology Unit at the University Hospital of Geneva (Geneva, Switzerland) and a member of the European Society of Cardiology (ESC) Committee for Practice Guidelines. The first case with this device was performed in the Hôpital du Valais (HVS, Sion, Switzerland).
The first patient was a 78-year-old male with a lesion in the left anterior descending (LAD) artery. A 2.5x12mm Rise SC balloon was used, and no complications were reported during the procedure.
Balloon angioplasty is mainly performed for the purpose of predilatation/lesion preparation prior to coronary stenting, followed by post-dilatation to optimise stent expansion. The goal of lesion preparation is to facilitate stent delivery and expansion. In some patients, direct stenting without predilatation can be performed, but predilatation is mandatory in severely stenotic lesions.
In most percutaneous coronary intervention (PCI) procedures, predilatation is performed using Semi-Compliant (SC) balloons at moderate pressure (8-10 atm) as they have a lower crossing profile than non-compliant (NC) balloons. The Rise SC balloon has been designed and developed to improve trackability and crossing profile and is aimed at having comparable performance to currently available “best-in-class” SC balloons, Biosensors said in a press release.