SQ Innovation has announced topline results from the SUBCUT HF II trial of its product Lasix ONYU, a novel subcutaneous formulation of furosemide for people with heart failure-associated oedema.
Patients randomised to treatment at home spent an average of four fewer days in the hospital compared to those treated with standard-of-care intravenous (IV) furosemide, according to the trial’s results, presented at Heart Failure 2026 (9 – 12 May, Barcelona, Spain), the annual congress of the Heart Failure Association (HFA) of the European Society of Cardiology (ESC).
SUBCUT HF II is a multicentre, open-label, randomised controlled trial conducted across 20 National Health Service (NHS) hospitals in the UK. The trial was co-sponsored by the University of Glasgow and the National Health Service (UK) Greater Glasgow and Clyde Health Board and led by principal investigators Ross T Campbell and Mark C Petrie of the Glasgow Cardiovascular Research Centre, University of Glasgow (Glasgow, Scotland).
“Oedema in heart failure is a leading reason for hospital admissions worldwide,” said Petrie. “This novel treatment option relies on the same therapeutic fundamentals that have been in place in the hospital since the 1960s, and enabling this to now be done at home by a patient or caregiver fundamentally changes the nature and trajectory of this very common and burdensome disorder.”
The trial evaluated at-home treatment of heart failure patients as an alternative to standard hospital treatment. For the study, 172 patients with heart failure were enrolled shortly after they were admitted to a hospital for treatment of oedema or fluid overload. Ninety-two patients were randomised to receive at-home treatment with Lasix ONYU and 80 received in-hospital treatment with IV diuretics.
Patients randomised in the study had a mean age of 71 years and were representative of a typical hospitalised heart failure population requiring diuretic therapy, with a median NT-proBNP of 3,365pg/mL, peripheral oedema present in 92%, and pleural effusion in 29%.
The primary endpoint was an avoidance of a minimum of 3.5 days of inpatient care also referred to as Days Alive and Out of Hospital (DAOH) over the 30-day period starting with the moment of randomisation. The primary endpoint reflects that the at-home therapy was effective and safe with low readmission due to treatment failure or admission for complications.
The study demonstrated four more days on its primary endpoint of DAOH at 30 days (p<0.001). Secondary endpoints included: length of index hospitalisation, change in health status at 60 days (assessed by Kansas City Cardiomyopathy Questionnaire [KCCQ-12]), days spent alive and out of hospital from randomisation to 60 days, total number of heart failure (HF) hospitalisations at 60 days, cardiovascular (CV) death or first HF hospitalisation at 60 days; CV death at 60 days, safety as determined by treatment emergent adverse events (TEAEs)
The researchers reported length of index hospitalisation reduced by 5.5 days (p<0.001), and benefits of DAOH remained at 60 days (4.23 DAOH difference between groups—p<0.001).
Secondary endpoints supported that aside from the avoidance of inpatient days, at-home Lasix ONYU treatment was equivalent to in-hospital treatment.
Patients in the at-home treatment arm received a full therapeutic dose by self- administration of approximately 11 units on average (880 mg). This total dose for an episode of oedema in heart failure is consistent with published literature for in-hospital treatment.
Adverse events were infrequent, and no device-related serious adverse event occurred. Lasix ONYU treatment was generally well tolerated.
“The SUBCUT HF II study represents an important milestone on this path home for the many patients for whom this is an option,” said Pieter Muntendam, founder, president and CEO of SQ Innovation. “This is the first outcomes study of home use of subcutaneous furosemide that meets the goals of demonstrating hospital avoidance and safe home use.”
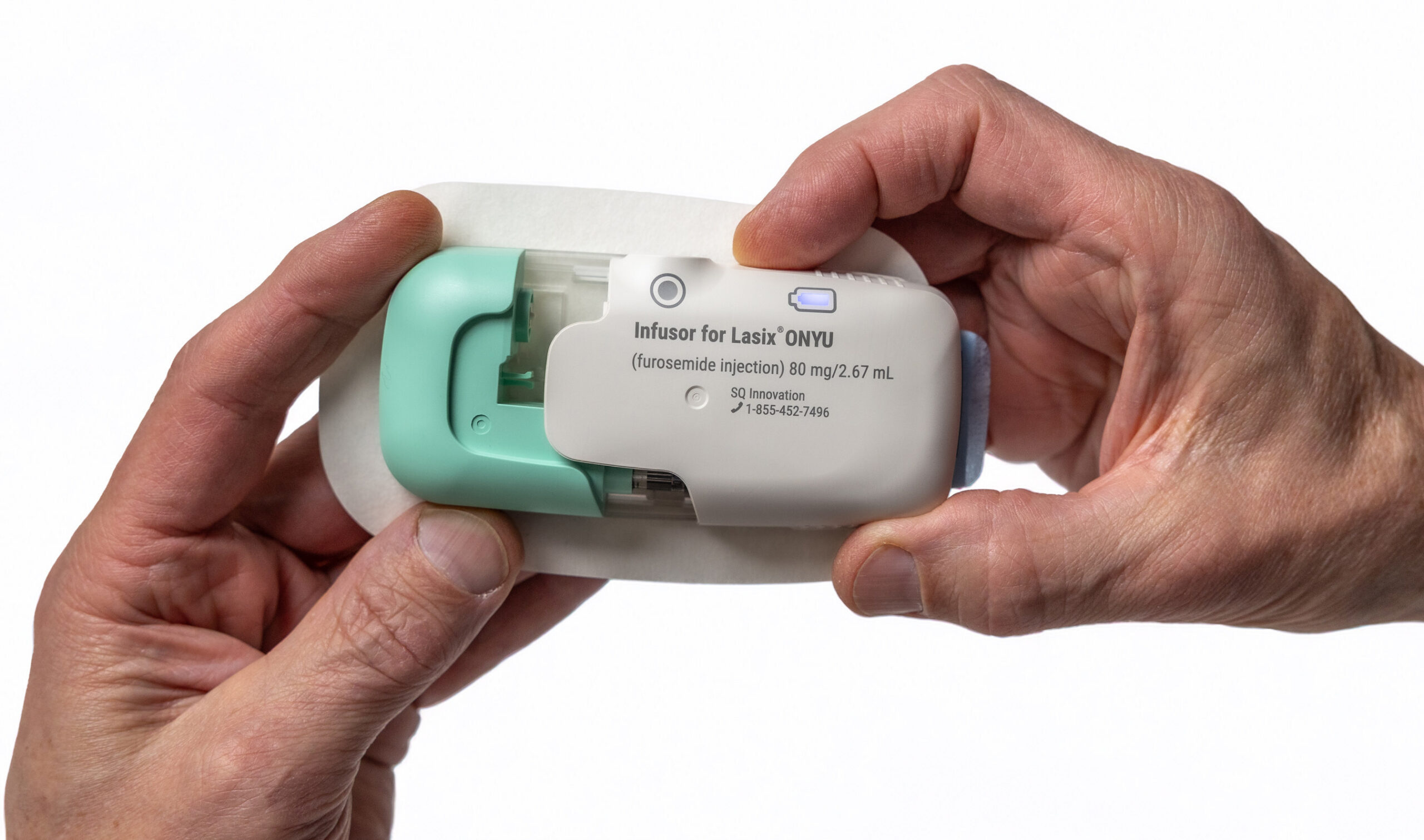
Lasix ONYU is a novel drug-device combination that was approved by the US Food and Drug Administration (FDA) in October 2025, for the treatment of oedema in adult patients with chronic heart failure. The Lasix ONYU Infusor administers 80mg of the diuretic furosemide over a period of five hours, resulting in significant diuresis similar to IV, but in a controlled manner designed for at-home administration to minimise risk of orthostatic hypotension and falls.