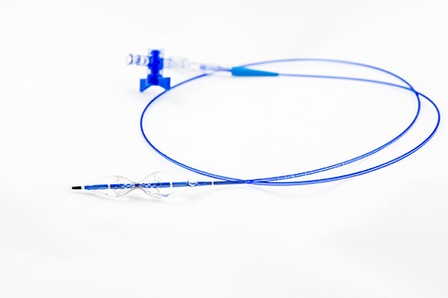
 Positive results of a single-centre, retrospective study using the Neovasc Reducer have been published in The Netherlands Heart Journal. The paper documents the results from 23 consecutive patients who were implanted with the Reducer in order to treat their severe refractory angina.
Positive results of a single-centre, retrospective study using the Neovasc Reducer have been published in The Netherlands Heart Journal. The paper documents the results from 23 consecutive patients who were implanted with the Reducer in order to treat their severe refractory angina.
The published results demonstrated that in all 23 patients the Reducer device was successfully implanted in a day hospital setting without any adverse events. After a median follow-up of nine months (range eight to 14 months) it was found that 17 of 23 patients showed at least one class improvement in their Canadian Cardiovascular Society (CCS) angina score: eight patients (34.8%) improved by one CCS class, seven (30.4%) by two CCS classes and two (8.7%) by three CCS classes.
“Despite the large range of available anti-ischaemic medical therapies and revascularisation techniques, a considerable number of patients still suffer from angina which is refractory to medical treatment,” says Pierfrancesco Agostoni, interventional cardiologist and senior author of the study. “The results from this real world cohort, albeit from one centre and a relatively small sample size, are consistent with previous studies using Reducer such as COSIRA and found Reducer implantation to be safe and efficacious, with the majority of patients demonstrating significant improvement in angina symptoms following treatment.”
In 2016 Neovasc initiated the REDUCER-I (An Observational Study of the Neovasc Reducer System), in Belgium, Germany, the Netherlands, Switzerland and the UK. The study is a 400-patient, 40-site, post-market, multicentre, multi-country, three-arm, five-year follow-up study. Its primary endpoint is the percentage of subjects who experience improvement in their angina symptoms defined as a reduction in CCS grade at six months as compared to baseline. This study is intended to provide clinical evidence supporting the use of the Reducer in this patient population.