December marked a significant milestone for a major study into the use of short duration dual anti-platelet therapy (DAPT) in high bleeding risk patients following stenting procedures, with patient recruitment reaching the half-way point.
The ambitious investigator-initiated MASTER DAPT (Management of patients post bioresorbable polymer Stent implantation with an abbreviated DAPT regimen) study just randomised patient number 2,150—half-way towards the target of 4,300. Patients join the study from more than 120 hospitals across 32 countries in Europe, Japan, Asia, Australia and Latin America.
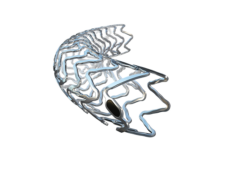
The study compares abbreviated versus prolonged DAPT, following implantation with Ultimaster or Ultimaster Tansei drug-eluting bioresorbable polymer stents, in ‘all-comer’ patients, presenting with high bleeding risk features. The study primary end-points are non-inferiority for net adverse clinical events; superiority for bleeding; and non-inferiority for ischaemic endpoints of abbreviated versus prolonged DAPT, at one year.
The Ultimaster DES has extensive real-world clinical data, having been studied in a population of over 40,000 patients. Both Ultimaster and the recently launched Ultimaster Tansei are designed to promote optimal vessel healing and therefore hypothesised to facilitate a shortened DAPT regimen. This hypothesis was confirmed in the DISCOVERY 1TO3 clinical trial that proved an excellent strut coverage of nearly 90% as early as 1 month.1 Robust safety data includes recently published results from the CENTURY II trial that showed a low stent thrombosis rate of 0.2% between 1 and 5 years.2
A shorter DAPT protocol will save substantial healthcare resources by reducing cost for DAPT, the number of hospitalizations for bleeding, and the number of working days lost.
References
- Smits P. Presented at EuroPCR 2015
- Wijns W. et al. EuroIntervention 2018;14:e343-51;5