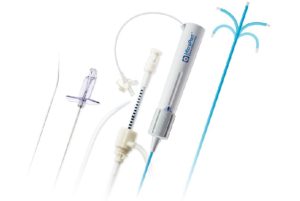
MicroPort has been given CE approval for its PathBuilder Steerable Introducer, PathBuilder Transseptal Guiding Introducer and PathBuilder Transseptal Needle.
A company statement outlines that the PathBuilder range of products helps to build a pathway from the femoral vein into atrium, and guide catheters to reach the target site. The products are available in different curve shapes, lengths, and diameters to allow access to different anatomic locations.
Among the portfolio is the PathBuilder Steerable Introducer, which the press rlease describes as offering a “stable and safe platform to reach the desired site with its bi-direction curve and braided shaft. The direction mark and zero-point mark on the handle are designed for precise control of the distal tip.”
Microport describes the approval of the PathBuilder products as laying a “solid groundwork for the company’s further exploration in the CE market”












