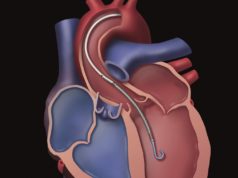
Abiomed has announced an expanded US Food and Drug Administration (FDA) approval for the Impella 5.0 and Impella LD heart pumps premarket approval (PMA) labelling in the treatment of cardiogenic shock, extending the duration of support for each pump from six days to 14 days.
The Impella 5.0 and the Impella LD are forward flow heart pumps that deliver up to five L/min, stabilising a patient’s haemodynamics, unloading the left ventricle, and perfusing the end organs, allowing for the potential of native heart recovery or a return to heart function baseline. The Impella 5.0 is implanted through the femoral or axillary artery and the Impella LD is implanted directly into the aorta. Both allow patients to walk around the unit while on support.
In a press release, Abiomed points out that Impella heart pumps have FDA PMA approval to treat heart attack patients in cardiogenic shock, and for shock associated with peripartum cardiomyopathy or myocarditis, and have the unique ability to enable the heart to rest and recover, allowing patients to return home with their own heart. The company says that the expanded indication allows for the opportunity to provide longer duration of support for critically ill patients and a longer period of assessment of heart recovery.
Jacob Abraham (Center for Advanced Heart Disease, Providence, USA) said: “Early recognition, escalation and a heart team approach are crucial for patients in cardiogenic shock. The Impella 5.0 and Impella LD’s ability to provide greater haemodynamic support and unload the left ventricle make them ideal tools for patients … who need longer duration support and will benefit from ambulation.”
The FDA’s original PMA approval indicating Impella as safe and effective for the treatment of cardiogenic shock was granted in 2016, and was based on an analysis of 415 patients from the FDA study RECOVER 1 and the US Impella registry, and an Impella literature review of 692 patients in 17 clinical studies. Additionally, more than 24,000 Impella patients supported by Impella devices were reviewed in a safety analysis.
According to the press release, the Impella heart pump platform is the most studied mechanical circulatory support device in the history of the FDA and has exclusive PMA approvals for high-risk percutaneous coronary intervention (PCI), as a therapy to allow for native heart recovery after cardiogenic shock derived from AMI or cardiomyopathy, and right ventricular heart failure.