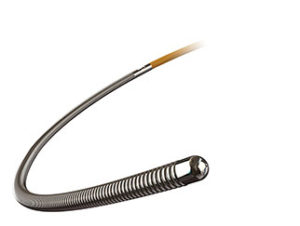
St Jude Medical has announced the FDA clearance and launch of its PressureWire X guidewire fractional flow reserve (FFR) measurement system. According to a press release, the latest generation of the PressureWire guidewire system is designed to offer improved “shapeability” and better shape retention with the aim of reducing vessel trauma. The press release notes that the guidewire provides the accuracy and simplicity that physicians require when treating patients during percutaneous coronary intervention (PCI), especially those with complex anatomies.
In addition to launching the latest PressureWire Guidewire, St Jude Medical has also launched the PRESSUREwire registry (Practical evaluation of FFR and its association alternate indices during routine clinical procedures). This is a multicentre clinical trial to determine the routine use of FFR measurement and clinical outcomes of FFR-guided PCI in patients with acute coronary syndrome.
The registry will build upon clinical trials designed to assess the positive outcomes associated with FFR technology, such as the St. Jude Medical sponsored FAME (FFR vs. angiography in multivessel evaluation) trials, which found that St Jude Medical PressureWire guidewire technology can improve patient outcomes over angiography alone in patients with coronary artery disease.
Annapoorna Kini, the director of the cardiac cath lab and the director of the interventional fellowship program at Mount Sinai Medical Center New York, USA, says: “For years, clinical research has confirmed that fractional flow reserve is one of the most important tools available when assessing coronary lesions and making informed treatment decisions during percutaneous coronary intervention The improved design and shape retention of the new PressureWire X Guidewire tip allows us to perform FFR measurements in more tortuous anatomies.”









