Atheart Medical has announced that it has received US Food and Drug Administration (FDA) approval for the start of the second phase of its ASCENT ASD US investigational device exemption (IDE) pivotal trial, evaluating the safety and efficacy of the reSept atrial septal defect (ASD) occluder.
The prospective, single-arm study will compare the device with established performance goals for previously FDA-approved transcatheter ASD occluders.
“I am pleased the first phase of enrolment in our pivotal trial progressed smoothly and according to plan, this is an exciting milestone for the company,” said Laurent Grandidier, CEO of Atheart Medical. “As we initiate the second phase, our team is focused on adding clinical sites across the USA and expanding internationally to include several enrolling sites in France. I commend the team’s diligence to further validating the safety and efficacy of the reSept ASD Occluder, a critical step in our journey to evolve septal closure and provide a better solution for patients.”
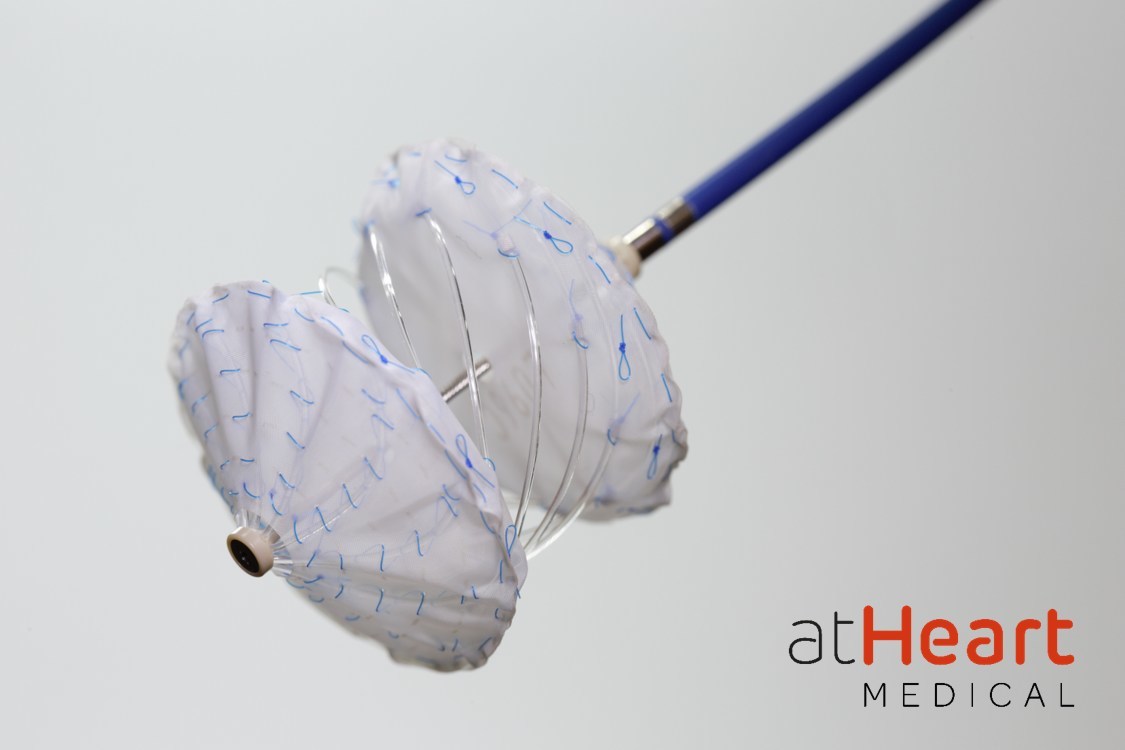
The reSept ASD Occluder aims to address the limitations of current occluders which have metallic frames that can place patients at risk of complications associated with long-term presence of metal in the heart and may limit future transseptal interventions, such as mitral valve interventions, Atheart said in a press release. Initial clinical experience demonstrates positive safety and performance in the closure of the ASDs treated with the company’s device, the release adds.
“The low-profile reSept Occluder is a dynamic system that allows for versatile physician control during the intervention, potentially adapting to the different patient anatomies to address ASDs,” commented Scott Lim, Professor of Medicine & Pediatrics at the University of Virginia in Charlottesville, USA, a leading enrolling site in the trial. “Over time, reSept’s metal-free frame resorbs, leaving a minimal implant behind. This is an exciting advancement that provides the potential to preserve future treatment options and potentially do better for our patients.”









