Findings from an interim analysis of PROTECT III have added to the evidence for improved outcomes with Impella heart pumps in protected percutaneous coronary intervention (PCI). PROTECT III is part of the PROTECT series of trials initiated by Abiomed in 2006, and follows on from PROTECT I, and the PROTECT II randomised controlled trial.
Published in 2012 by William O’Neill et al, PROTECT II randomised patients 1:1 to an intra-aortic balloon pump (IABP) and PCI or Impella 2.5® circulatory support device and PCI.1 It showed that use of an Impella leads to better outcomes for Protected PCI than an IABP in patients with reduced ventricular function. Based on the findings from the three PROTECT studies, as well as registry data, the US Food and Drug Administration granted Impella its highest level of safety and efficacy approval, a post-market approval (PMA) for a first-of-its-kind indication for high-risk PCI. Impella is included in eight clinical guidelines and more than 650 peer-reviewed publications.
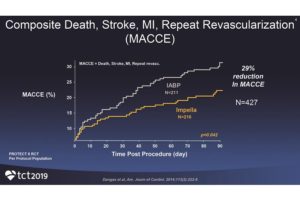
Interim findings from the prospective study PROTECT III, announced recently at the Transcatheter Cardiovascular Therapeutics scientific symposium (TCT 2019; 25–29 September, San Francisco, USA), demonstrated a reduction in the primary endpoint of death, stroke, myocardial infarction and repeat procedures at 90 days (major adverse cardiac and cerebrovascular events, MACCE) with Impella supported PCI, compared to PROTECT II (In PROTECT II: IABP 31%, n=210, vs. Impella 21.9%, n=215, p=0.033; In PROTECT III: Impella 16.8%, n=469, p<0.0001). The data were presented by Jeffrey J Popma, MD (director of interventional cardiology clinical services at Beth Israel Deaconess Medical Center, and professor of medicine at Harvard Medical School, Boston, USA).

The encouraging findings, he noted, were despite the fact that: “Compared to PROTECT II, patients are older [average of 71 years], more often women [26%], and there were less Caucasians over time [33% non-Caucasians].”
In addition, Popma pointed out: “We thought we were aggressive in PROTECT II, but, in PROTECT III, the number of vessels treated was higher and the number of three vessels treated doubled (from 14% to 29%). The amount of atherectomy used was 40% in PROTECT III, whereas it was only 14% in PROTECT II. Despite this, the contrast volume in PROTECT III was lowered. The length of support was longer in PROTECT III, in part due to the fact that patients were sicker compared with PROTECT II.
Why provide left ventricular support during PCI?
As coronary interventional techniques progress and health care delivery improves, PCI has been extended to patients who would previously have been deemed untreatable, particularly those with reduced ventricular function. In addition, advancing age and increased comorbidities have put more patients at prohibitive risk for coronary artery bypass grafting, rendering high-risk PCI as the sole revascularisation therapy for this population. While there is no absolute definition of a high-risk intervention, it is generally considered in those patients with reduced left ventricular systolic function, complex coronary anatomy, or advanced co-morbidities, frailties, or disabilities, including end-organ dysfunction (i.e., kidney disease or lung disease). Mechanical circulatory support in PCI may be used as an adjunct in such cases, with the goal of providing sufficient forward cardiac output to maintain myocardial flow and end-organ perfusion and to reduce left ventricular volume, wall stress, and end diastolic pressure thereby improving coronary flow) during the procedure. Use of an appropriate left ventricular support device prior to initiating a high-risk coronary intervention allows time to safely perform an optimal coronary intervention in patients with advanced coronary artery disease to allow more extensive revascularisation during the procedure. Ventricular support enables the operator to proceed confidently without the risk of hemodynamic collapse that would require the subsequent for emergency bailout use of other advance support devices, such as an intra-aortic balloon pump or extracorporeal support.

How does Impella work?
Impella 2.5 and Impella CP® heart pumps function by pulling blood from the left ventricle through an inlet area near the tip and expelling it from the catheter into the ascending aorta. The device can be inserted using a standard catheterisation procedure through the femoral artery, into the ascending aorta, across the valve and into the left ventricle. Impella CP with SmartAssist® is one of the latest innovations on the Impella platform, integrating the performance of the Impella CP heart pump with state-of-the-art SmartAssist technology. It uses real-time intelligence to optimise positioning and management of the device, and new haemodynamic sensor technology allows it to be repositioned in the ICU without the need for imaging (for ventricularised pumps only); the optical sensor senses aortic pressure while the micro-axial motor senses pressure between the aorta and the left ventricle. It has been designed with a simplified set-up, for improved ease of use and faster set-up time.
Expanded post-approval study of protected PCI
PROTECT III is an ongoing, prospective, single arm FDA post-approval study for the premarket approval (PMA) of Impella 2.5 and Impella CP heart pumps in high-risk PCI, and is the largest FDA study of haemodynamically supported high-risk PCI patients. It is being conducted at 45 hospitals in the USA, with 898 patients enrolled between March 2017 and July 2019—571 to Impella CP and 327 to Impella 2.5—all of whom have a high-risk PCI indication.
Popma stressed: “We are not asking the question: ‘Is CABG better than PCI with normal left ventricular function and stable angina?’ We are saying: ‘In those patients who are high risk, what is the appropriate method of support for advanced and complete revascularisation?’”
Popma stated that comparison of revascularisation strategies by surgical risk category in recent revascularisation trials found that studies such as SYNTAX, EXCEL, NOBLE and BCIS enrolled patients at low- and medium-risk surgical risk, whereas the highest surgical risk patients were enrolled in PROTECT II and PROTECT III, most of whom were not candidates for surgery at all, in addition to high anatomical complexity.
“These are the challenging patients in our clinical practices who have few alternatives. The PROTECT studies addressed these patients,” Popma explained: “The prospective PROTECT III clinical study is collecting data for Impella use with the same rigour as a pre-Market FDA study, including the use of core laboratories, clinical site monitoring, and clinical events committees. We are already now at 898 patients in the PROTECT III post-approval study, and that is going to continue to grow. Now we are going to be able to do some analyses on thousands of patients … and have the ability to look a little bit deeper on the clinical outcomes.”
 Clinical importance of complete revascularisation to reduce late events
Clinical importance of complete revascularisation to reduce late events
A key aspect demonstrated by both PROTECT II and PROTECT III is the compelling evidence on complete revascularisation, with the benefits of Impella more pronounced with extensive revascularisation.
Crucially, said Popma: “Use of the Impella with extensive revascularisation prevented late adverse events, including repeat revascularization, rather than in-hospital events, but late adverse events are critically important to physicians and their patients.”
PROTECT II had two primary endpoints—a 30-day primary endpoint and a 90-day composite endpoint of major adverse events, including death, myocardial infarction, stroke/transient ischaemic attack (TIA) and repeat revascularisation. In PROTECT II, the 30-day incidence of major adverse events was not different for patients with IABP or Impella 2.5 haemodynamic support. However, improved outcomes were observed at 90-days for Impella 2.5-supported patients.
Popma highlighted the divergence: “From the time the patient is discharged to 90 days later [the cumulative] event rates continue to separate. Reduction in out of hospital events is where the benefit is seen.”
The clinical benefit of Impella Protected PCI was a reduction of major adverse events post-discharge, accompanied by an improvement in ventricular function and heart failure symptoms. On the endpoint of death, stroke, MI and repeat revascularisation, event rates were 18% with IABP and 10% with Impella (p=0.01), and for death, stroke and MI they were 13% and 7% (p=0.047), respectively. These post-discharge clinical benefits were more pronounced in patients who underwent more complete revascularisation with Impella support.
In patients with limited revascularisation there was little difference between patients treated with an Impella heart pump and balloon pump in MACCE (32.2% IABP vs. 35.1% Impella) whereas in complete revascularisation MACCE events were 29.3% for IABP vs. 15% for Impella.
Popma summed up: “Impella maintains procedural haemodynamics, allowing for more complete revascularisation. In those patients who had extensive revascularisation, there was a profound difference in outcomes compared with patients treated with an intra-aortic balloon pump.”
This finding is supported by findings from the Roma-Verona registry in 2019 (Burzotta et al), which looked at the use of Impella in protected high-risk PCI and found that more complete revascularisation was associated with significant left ventricular ejection fraction (LVEF) improvement and survival.2
The finding of less required haemodynamic support after PCI with Impella was a “very provocative” observation in PROTECT II, Popma stated; total support time following IABP-PCI was 8.23±21 hours vs. 1.86±2.7 hours with Impella (p<0.001). And 37.7% of IABP patients were discharged from the cath lab on the balloon pump vs. 5.6% of patients treated with Impella (p<0.001).
“After the procedure, for whatever reason, the clinician needed to maintain the balloon pump for a while. Usually that is because the patient has a soft blood pressure. And a lot of patients went directly up to the unit with the balloon pump in place, so they ultimately required longer haemodynamic support.”
In addition, PROTECT II found half as many hypotensive events (defined as a mean arterial pressure of <65mmHg) per patient with Protected PCI: IABP 0.96 vs. Impella 0.45, a reduction of 53% (p=0.001).
Impella 2.5 provided superior haemodynamic support in comparison with IABP, with maximal decrease in cardiac power output from baseline of -0.04±0.24W in comparison with -0.14±0.27W for IABP (P=0.001).1 “And that is what observe in the catheterisation lab when we just don’t see the patients get hypotensive as we perform extensive revascularisation,” said Popma.
Impella and acute kidney injury
PROTECT III is also taking a closer look at the development of acute kidney injury (AKI). High-risk PCI patients face elevated risk of AKI due to high levels of contrast and long procedures. In addition, chronic kidney disease is a common comorbidity. A substudy of PROTECT III evaluated 106 patients for AKI rate and compared them to a propensity matched control group (also 106 patients).
“Patients with AKI are at greater risk of adverse events. Low ejection fraction and baseline haemodynamic compromise may result in renal hypoperfusion,” said Popma.
The PROTECT III substudy cohort consisted of patients with recorded baseline creatinine, and at least one follow-up creatinine measurement within 48–72 hours of PCI; patients on dialysis at the time of PCI were excluded. In the control group were elective or urgent high-risk PCI patients treated without Impella support in the AKI study performed at the University of Louisville.3,4 Patients were matched 1:1 based on age, gender, diabetes, contrast volume, and glomerular filtration rate (GFR).
The analysis demonstrated an AKI rate of 24.5% in patients with no support vs. 5.7% in the PROTECT III cohort (p=0.0002). In addition, the rate of severity was lower in the Impella-protected patients, with respective values of 9.4% vs. 5.7% for severity of AKI-1, 10.4% vs.0% for AKI-2, and 4.7% vs. 0% for AKI-3 in unprotected patients and those in PROTECT III.
Where to next for Impella heart pumps? Popma surmised: “We have learned a lot about PCI with left ventricular support over the last several years; we have shown the benefit of haemodynamic support and the importance of extensive revascularisation; we have learned that these are late events that are prevented by a more effective initial procedure. It really sets the stage for what we could do next. And what we could do next is use the left ventricular support to achieve complete revascularisation, and ultimately evaluate the use of contemporary “best practice” methods for Impella support in patients who are high risk for surgery.”
REFERENCES:
- W O’Neill et al. Circulation 2012. doi.org/10.1161/CIRCULATIONAHA.112.098194
- F Burzotta et al. Journal of Interventional Cardiology 2019. doi.org/10.1155/2019/5243913
- MP Flaherty et al. Catherization and Cardiovascular Interventions, 2019. doi.org/10.1002/ccd.28400
- MP Flaherty et al. Circulation Research 2017. doi.org/10.1161/CIRCRESAHA.116.309738