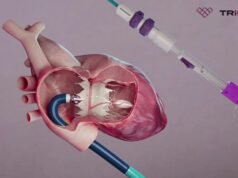
TruLeaf Medical is to conduct a first-in-human clinical trial of a “two-stage” transcatheter mitral valve replacement (TMVR) procedure, it has been announced.
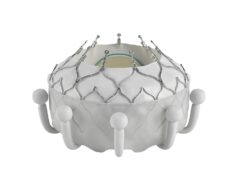
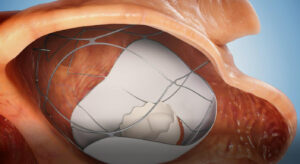
The trial will involve the use of the company’s RoseDoc platform, described as a docking station that is implanted in the left atrium, which precedes the implantation of an artificial biological mitral valve prosthesis several weeks later. The two-stage procedure is intended to achieve optimal anchoring of the valve prosthesis, a press release from AllMeD Solutions, of which Truleaf Medical is a subsidiary, claims.
Benjamin Spencer, TruLeaf Medical CEO, said: “The main challenge with existing TMVR technologies is achieving optimal anchoring of the valve prosthesis to the heart, given the complex native mitral valve anatomy and physiology. The RoseDoc TMVR platform is technically simple, safe and has been proven to be effective in rigorous long-term animal testing.
“Complete elimination of the leak prevents progressive dilation of the heart, which, by itself, in a vicious cycle, worsens the leak, leading to a progressive weakening of the heart muscle and intractable heart failure,” Benjamin Spencer, CEO of Truleaf Medical, said, “At present, patients with severe mitral valve leak refractory to maximal medical treatment remain without an effective treatment. The vast majority of these patients are declined surgery due to prohibitive risk. The RoseDoc unique TMVR platform literally gives these patients new hope.”
The final design of the TMVR platform was tested by Horst Sievert (CardioVascular Center, Frankfurt, Germany), who has recently joined TruLeaf’s Scientific Advisory Board and will lead the human trial.
In the press release, Sievert was quoted as saying: “The concept of the product is ingeniously simple and very promising. I look forward to leading the first human trial of this innovative technology.”