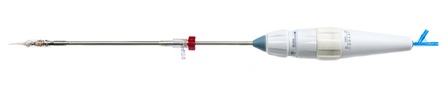
Transcatheter Technologies GmbH, a medical device company that has developed a full range of transcatheter valve implantation systems for catheter-based heart valve therapy, has sold its technology portfolio to Venus Medtech. A press release reports that the Transcatheter Technologies’ technology will be the basis of a next-generation product portfolio developed and marketed by Venus Medtech for the Chinese and international markets.
According to the press release, under the agreement, Transcatheter Technologies’ co-founders cheif executive officer and cardiac surgeon Wolfgang Goetz and chief technology officer and engineer Hou-Sen Lim will assist Venus Medtech with the transition and commercialisation of the acquired technology for up to three months.
Venus Medtech develops and manufactures an advanced interventional artificial cardiac valve system. It is a leading heart valve developer in China, and its transcatheter aortic valve has completed clinical trials and follow up, pending Chinese FDA approval. Its transcatheter pulmonic valve is completing its clinical trials. Both of the valves are expected to be the first transcatheter valves approved in China. The company are also developing the next generation heart valves with pre-crimping technology. Venus Medtech sets its goal as a worldwide transcatheter heart valve company.
Goetz says: “Transcatheter Technologies was founded with the goal of making non-invasive heart valve implantation safer for patients, therefore expanding the number of patients who may benefit from this procedure. This agreement with Venus Medtech helps us continue this mission and will fuel the continued development of our next-generation technology platform for the treatment of various structural heart diseases in the Chinese market. Our third-generation transcatheter aortic valve implantation (TAVI) system—Trinity—has already completed two-year follow-up for a pilot study of its Trinity TAVI system that is designed to be the world’s first ‘truly repositionable ’TAVI system.”










