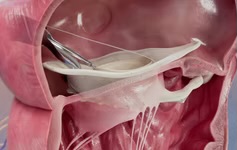
Rebecca Hahn is the principal investigator of the SCOUT (the early feasibility of the Mitralign percutaneous tricuspid valve annuloplasty system in patients with chronic functional tricuspid regurgitation) trial, which is evaluating the use of Mitralign’s transcatheter tricuspid repair system for the management of tricuspid regurgitation. She reviews the potential role of the Mitralign system in treating these patients.
Functional or secondary tricuspid regurgitation is the most common aetiology of severe tricuspid regurgitation in the western world.1 Interest in the tricuspid valve has increased in recent years2–4 with the recognition of the impact of secondary tricuspid regurgitation on outcomes.5–10 The prevalence of secondary tricuspid regurgitation with mitral regurgitant disease is more than 30%2,3 with some studies suggesting more than 1.6 million patients in the US may currently have this condition.11 Current guidelines suggest a transthoracic tricuspid annular dimension in the apical four-chamber view of >40mm or >21mm/m2 indicates significant annular dilation and an increased risk of persistent or progressive tricuspid regurgitation after isolated mitral valve surgery.12 The only Class I indication for surgical tricuspid valve repair is with concomitant left heart valve surgery. However despite studies clearly showing the lower risk of tricuspid valve repair or replacement using this approach,13,14 its adoption has not been universal and a significant population of patients with symptomatic severe tricuspid regurgitation following mitral valve surgery, are faced with excessive surgical risk for re-operation. In patients who develop severe tricuspid regurgitation late after left heart valve surgery, operative mortality may be as high as 35%.2,15–18 Additionally, recent studies of both post-transcatheter aortic valve replacement patients and post-MitraClip (Abbott Vascular) patients have shown higher mortality in patients with significant tricuspid regurgitation.19,20 Thus the patient population with high risk, severe, symptomatic tricuspid regurgitation may actually continue to expand, increasing the need for percutaneous solutions.
The Mitralign system (Mitralign) has completed its mitral CE mark trial in Europe and recently reported their first-in-human implantation of their system on the tricuspid annulus.21 It places two connected pledgeted sutures within the tricuspid valve annulus by means of a trans-jugular venous approach. Once the pledgets have been delivered within the annulus at pre-defined locations and distance apart, a dedicated plication lock device is used to bring the two pledgeted sutures together, plicating the annulus by at least 2cm and effectively bicuspidising the tricuspid valve. This device replicates a well-established surgical bicuspidisation repair approach.22 An essential component of successful placement of the sutures is intraprocedural imaging, currently using transoesophageal echocardiography (TEE)—which permits imaging with higher frequencies for improved spatial resolution and a larger number of windows for a more comprehensive examination of the entire tricuspid valve apparatus. Standardisation of the both two-dimensional and three-dimensional echocardiographic image acquisition and display for the tricuspid valve23,24 has advanced the field immeasurably, improving communication with the interventionalist.
In a series of compassionate use patients in Europe, a two-pledget approach successfully reduced annular area by 41%, and tricuspid effective regurgitant orifice area by 62% with no procedural complications (unpublished data). Based on this early success, the SCOUT trial will start enrolling by the end of 2015. This trial will enrol up to 15 patients to assess the early safety and feasibility of the system for the treatment of patients with functional tricuspid regurgitation (minimum requirement is moderate tricuspid regurgitation) in whom left-sided valve or cardiac surgery is not planned. Although the primary outcome is safety and feasibility, this initial trial should enhance our understanding of not only the appropriate patient population for treatment, but also the generalisability of the imaging and implantation technique.
References
- Cohen et al. J Thorac Cardiovasc Surg 1987; 94(4) :488–97.
- Antunes et al. Heart 2007; 3(2): 271–76.
- Rogers et al. Circulation 2009; 119(20): 2718–25.
- Shah et al. Curr Probl Cardiol 2008; 33(2): 47–84.
- Nath et al. J Am Coll Cardiol 2004; 43(3): 405–09.
- Voelkel et al. Circulation 2006; 114(17): 1883–91.
- Bustamante-Labarta et al. J Am Soc Echocardiogr 2002; 15(10 Pt 2): 1160–64.
- Hung et al. Am J Cardiol 1998; 82(10): 1301–03
- Agricola et al. Eur J Heart Fail 2012; 14(8): 902–08.
- Agricola et al. J Cardiovasc Med (Hagerstown) 2015. Epub
- Stuge et al. The Journal of Thoracic and Cardiovascular Surgery 2006; 132(6): 1258–61.
- Nishimura et al. Journal of the American College of Cardiology 2014; 63(22):2438–88.
- Calafiore et al. The Annals of Thoracic Surgery 2009; 87(3): 698–703.
- Chikwe et al. J Am Coll Cardiol 2015. Epub
- Hornick et al. The Journal of Heart Valve Disease 1996 ;5(1): 20–25.
- Mangoni et al. European Journal of Cardio-Thoracic Surgery; 2001; 19(1): 68–73.
- Kwon et al. The American Journal of Cardiology 2006; 98(5): 659–61.
- Bernal et al. The Journal of Thoracic and Cardiovascular Surgery 2005; 130(2): 498–503.
- Lindman et al. Cardiovascular Interventions 2015; 8(4).
- Ohno et al. Eur Heart J Cardiovasc Imaging 2014; 15(11): 1246–55.
- Schofer et al. Journal of the American College of Cardiology 2015. Epub
- Navia et al. The Journal of Thoracic and Cardiovascular Surgery 2010; 139(6): 1473–82
- Lang et al. Eur Heart J Cardiovasc Imaging 2012; 13(1): 1–-46.
- Hahn et al. J Am Soc Echocardiogr 2013; 26(9): 921–64.
Rebecca Hahn is director of Interventional Echocardiography, and associate professor of Medicine at Columbia University Medical Center/New York-Presbyterian Hospital, New York, USA