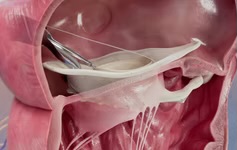
BioVentrix has announced the first-in-man use of its Revivent-TC Myocardial Anchoring System, via Less Invasive Ventricular Enhancement (the LIVE procedure). The procedure was performed by Gintaras Kalinauskas and Gierdrius Davidavicius (both of Vilnius University Hospital in Vilnius, Lithuania).
The successful procedure, used to reshape and reduce the left ventricle, was performed on a 61-year-old man suffering from advanced heart failure in the interventional catheterisation laboratory.
“Additionally, because of the much less invasive nature of the LIVE procedure, he was discharged just five days postoperatively, which will significantly decrease the cost associated with this treatment. With conventional surgical ventricular reconstruction, the duration of a typical hospital stay can range from six days to more than two weeks.”
“Before the LIVE procedure, this patient’s quality of life was severely impacted by his heart failure to the point where he could no longer walk even short distances without discomfort,” says Davidavicius, interventional cardiologist and co-investigator. “For patients this fragile, the risks and extended recovery of surgery often preclude any meaningful intervention. The LIVE procedure, which eliminates the most invasive elements associated with conventional surgical reconstruction, greatly expands the patient population that will be able to benefit from this life-saving procedure.”
The Revivent-TC System utilises a myocardial anchor identical the company’s Revivent Myocardial Anchoring System, which received CE mark in December 2012 and is commercially available and in clinical use at heart centres in all major Western and Eastern European markets.
The company also recently announced publication of clinical results in the European Journal of Cardio-Thoracic Surgery demonstrating the durability of the Revivent System. In that study, patients sustained a mean reduction in left ventricular end-systolic volume index (LVESVI), the key prognostic indicator for HF, to 43.9 mL/m2, well below the 60mL/m2 threshold recently defined as providing a survival benefit in heart failure patients. These clinical results are expected to continue with the new transcatheter delivery approach of the Revivent-TC System.
Prior to the Revivent and Revivent-TC Systems, reshaping of the left ventricle involved an invasive procedure (surgical ventricular restoration), which required cutting through the patient’s sternum to expose the heart, then stopping and supporting it with cardiopulmonary bypass while incisions into the heart muscle were made to remove the scarred, non-functioning tissue. The invasive nature of this procedure limits the use of this procedure due to the fragile nature of many heart failure patients the company states. In contrast, the LIVE procedure is performed without the need of cardiopulmonary bypass or making incisions into the heart, and now the Revivent-TC System also removes the need to open the patient’s chest.
“There were no complications, and the patient’s heart function improved immediately and has continued to do so,” continues Davidavicius. “Additionally, because of the much less invasive nature of the LIVE procedure, he was discharged just five days post-operatively, which will significantly decrease the cost associated with this treatment. With conventional surgical ventricular restoration, the duration of a typical hospital stay can range from six days to more than two weeks.”
“We are very pleased with the outcome of this first-in-man LIVE procedure with the Revivent-TC System by the heart failure team in Lithuania,” said Kenneth Miller, president and chief executive officer of BioVentrix.