Shockwave Medical has announced the completion of its previously announced acquisition of Neovasc.
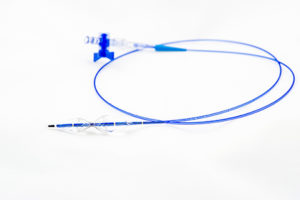
The Neovasc Reducer system is a first-of-its-kind technology to address refractory angina. Refractory angina is a chronic condition in which a patient suffers chest pain that cannot be controlled by conventional therapies.
It is estimated that each year, in the USA and the European Union (EU) alone, up to 300,000 new patients with obstructive coronary disease who are ineligible for conventional revascularisation experience refractory angina, despite guideline-directed medical therapy.
In addition, it is estimated that up to another 500,000 new patients present with angina and non-obstructive coronary artery disease in the USA and the EU each year.
The Reducer has been granted Breakthrough Device designation by the US Food and Drug Administration (FDA), is CE-marked and is currently enrolling patients in the COSIRA-II study, a randomised clinical trial being conducted under an investigation device exemption (IDE) intended to support FDA approval for patients with coronary obstructive refractory angina.
Shockwave has acquired all of the outstanding common shares of Neovasc upfront by way of a statutory plan of arrangement.
As a result of the completion of the Arrangement, Neovasc’s Common Shares will be delisted from the Toronto Stock Exchange. Neovasc has also requested that the Nasdaq Stock Market LLC file a delisting application on Form 25 to report the delisting of the Common Shares of Neovasc from Nasdaq.










