Procyrion has announced the enrolment of the first patients in the company’s investigational device exemption (IDE) pivotal trial.
The DRAIN-HF study will evaluate the Aortix percutaneous mechanical circulatory support (pMCS) technology in patients with acute decompensated heart failure (ADHF) who are unresponsive to standard medical therapy. The first patient in the randomised portion of the trial was enrolled by Bassem Chehab (Ascension Via Christi Hospital, Manhattan, USA).
“I am thrilled to enrol the first patient in this important trial. Today, there are no effective treatment options for patients with ADHF who remain congested despite being treated with high dose intravenous diuretic therapy. These patients have very poor outcomes in terms of death and rehospitalisation and therefore new therapy options are critically needed,” stated Chehab.
The study also includes a registry for advanced heart failure patients who are end stage and not considered to be surgical candidates for a heart transplant or left ventricular assist device (LVAD) implant due to excessive fluid retention and poor kidney function. The first patient in the registry was enrolled at Henry Ford Hospital (Detroit, USA) by Gillian Grafton.
“The Aortix pump demonstrated promising results to help off-load fluid and improve kidney function. I am excited to be able to include the advanced heart failure patients in the trial,” commented Grafton.
The DRAIN-HF pivotal trial is evaluating the safety and effectiveness of the Aortix pump compared to standard of care medical therapy including high dose intravenous (IV) diuretics in patients hospitalised with ADHF who have persistent congestion and demonstrated diuretic resistance despite optimal medical management.
The study will enrol up to 268 patients across 45 centres in the USA and will include a randomised arm as well as a registry arm that will enrol patients who are targeted for a heart transplant or left ventricular assist device (LVAD) but are too sick to be considered for surgery. The company recently announced it has received Centers for Medicare & Medicaid Services (CMS) approval for Medicare coverage during the DRAIN-HF pivotal trial.
“The initiation of the DRAIN-HF pivotal study represents a major milestone in the company’s commitment to make Aortix available for these acutely decompensated heart failure patients who have failed aggressive standard of care medical therapies,” said Eric S Fain, president and chief executive officer, Procyrion. “We look forward to working with our investigators and research teams to advance the scientific evidence around this groundbreaking technology.”
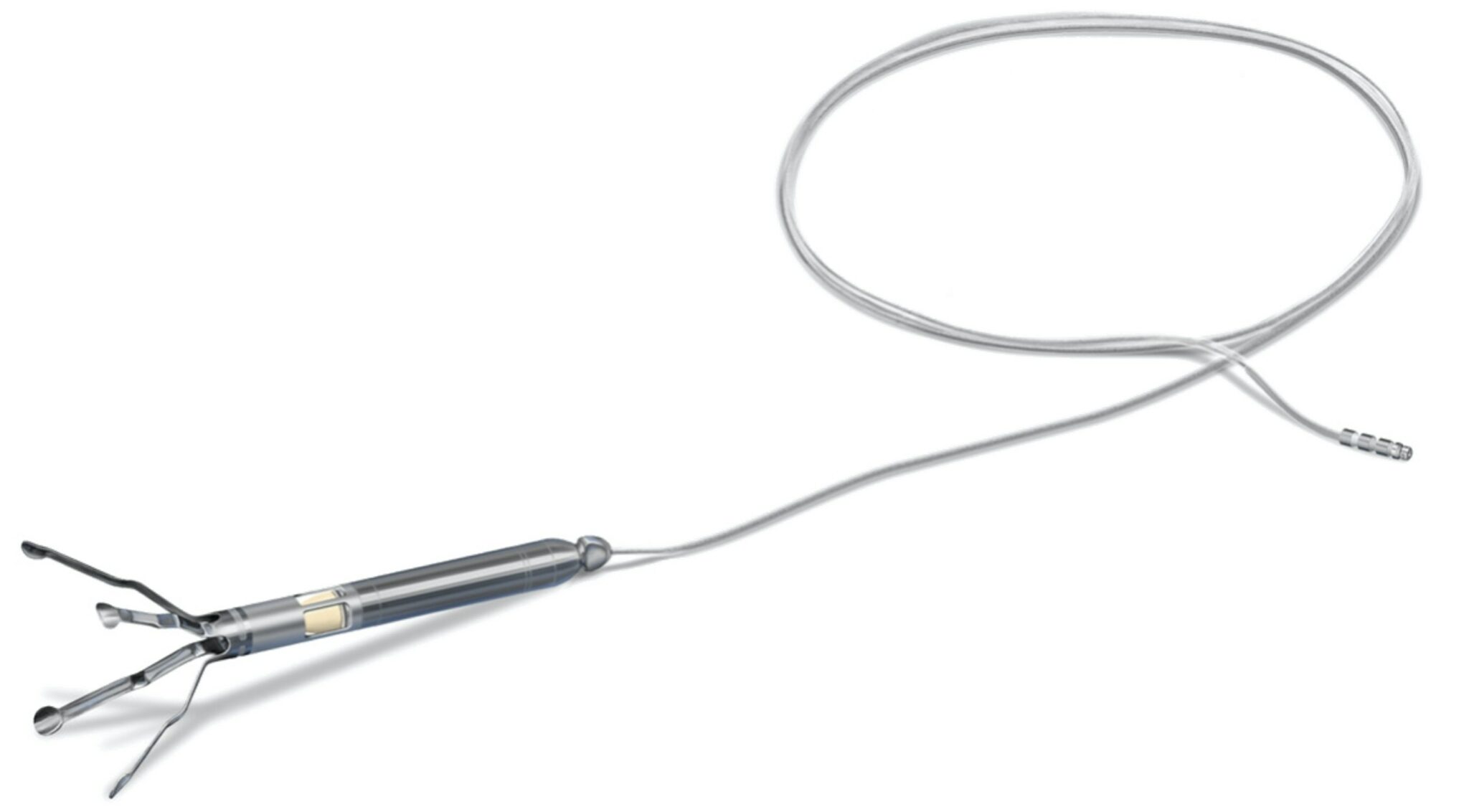
Aortix is a catheter-deployed pump that is placed in the descending thoracic aorta and designed to directly unload the heart and improve cardiac performance while increasing perfusion to the kidneys.
In the CRS Pilot Study, which was published in the Journal of the American College of Cardiology: Heart Failure, patients demonstrated rapid decongestion with substantial removal of excess fluid and improved haemodynamics, as well as significant improvements in kidney function, cardiac function, and patient-reported assessment of shortness of breath sustained out to 30 days after treatment with the Aortix pump.













