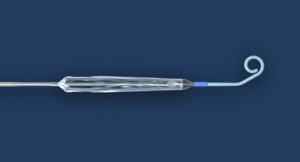
The US Food and Drug Administration (FDA) has granted breakthrough device designation to Abiomed’s Impella ECP expandable percutaneous heart pump. The designation means the FDA will prioritise Impella ECP’s regulatory review processes including design iterations, clinical study protocols and pre-market approval (PMA) application.
Impella ECP is the smallest heart pump in the world and the first to be compatible with small bore access and closure techniques. It measures 9 French (3mm) in diameter upon insertion and removal from the body. While in the heart, it expands to support the heart’s pumping function, providing flow greater than 3.5L/min.
The FDA granted breakthrough device designation in part based on positive clinical data from the first 21 Impella ECP patients treated as part of an FDA regulated early feasibility study. In granting the designation, the FDA determined Impella ECP meets the FDA’s stringent requirements for a breakthrough device.
“This is yet another validation from the FDA of the clinical benefits of Impella technology and an affirmation of the innovative nature of Impella ECP which, due to its smaller vascular access size, has the potential to provide even safer procedures and be available to more patients who need hemodynamic support for coronary revascularisation,” said Chuck Simonton, Abiomed’s chief medical officer.
Impella ECP is an investigational device, limited by federal law to investigational use only.










