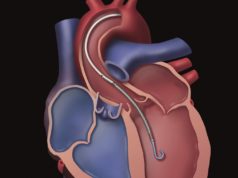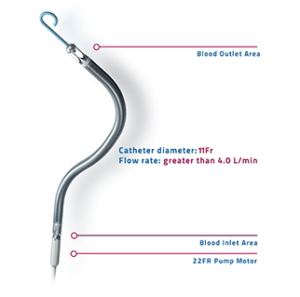
Abiomed has received FDA pre-market approval for its Impella RP heart pump. Culminating from five years of research, according to a press release, this approval follows the prior FDA hmanitarian device exemption (HDE) received in January 2015 and adds the Impella RP heart pump to Abiomed’s platform of PMA-approved devices.
The Impella RP system is indicated for providing temporary right ventricular support for up to 14 days in patients with a body surface area ≥1.5m2, who develop acute right heart failure or decompensation following left ventricular assist device implantation, myocardial infarction, heart transplant, or open-heart surgery.
Data submitted to the FDA in support of the Impella RP heart pump PMA included the RECOVER RIGHT FDA-approved, prospective, multicentre, single-arm study, which commenced after the company received FDA investigational device exemption (IDE) approval in November 2012 and concluded in 2014. Since that time, Abiomed completed a continuous access protocol (CAP) as well as a prospective, multicentre post-approval study.
Mark B Anderson (Hackensack University Medical Group, Hackensack, USA), who was the co-principal investigator for the RECOVER RIGHT trial, says: “The Impella RP has opened up a new era in cardiovascular medicine. With its percutaneous, single vascular access, the Impella RP offers physicians a minimally-invasive procedure for patients who have previously had limited options for treatment of right heart failure. The cumulative data from the FDA studies demonstrate that Impella RP potentially offers survival benefits for these critically ill patients in need of haemodynamic support.”
The Impella RP heart pump is reimbursed by Medicare and other major payers. Abiomed will complete a PMA post-approval study with 60 consecutive patients in Abiomed’s Institutional Review Board (IRB)-approved, FDA audited prospective cVAD Registry.
With this approval, the press release reports, the Impella RP heart pump is the only percutaneous temporary ventricular support device that is FDA-approved as safe and effective for right heart failure.