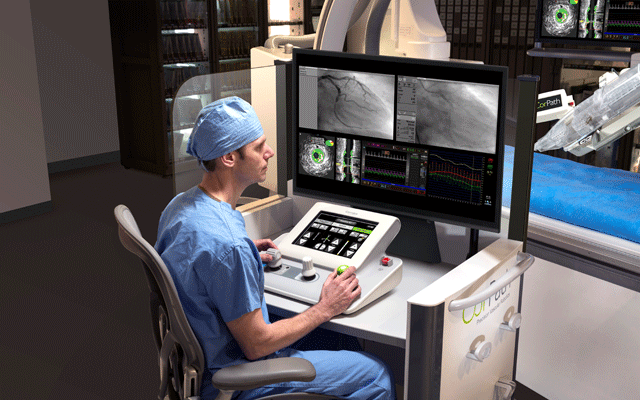
Corindus Vascular Robotics has received Pharmaceutical and Medical Device Agency (PMDA) approval for commercialisation of its CorPath GRX system in Japan. A press release notes that Japan is one of the largest markets in the world for percutaneous coronary interventions (PCI) with approximately 250,0001 PCI procedures performed annually.
It adds that the company is actively pursuing reimbursement for robotic PCI through the Japanese insurance system and plans a postmarket study of CorPath GRX. In early 2017, Corindus announced a strategic relationship with Japan Medicalnext (a wholly-owned entity of MC Healthcare; subsidiary of Mitsubishi Corporation) to become the exclusive distributor of Corindus products in Japan—bringing robotic precision and control to the large and growing PCI segment of the Japanese healthcare market.
Takafumi Ueno (Center of Cardiovascular Disease, Kurume University Hospital, Kurume, Japan) says: “I look forward to offering CorPath GRX to my patients during PCI, especially in complex procedures where precision and procedural control are extremely valuable. We are pleased to be one of the first Asia-Pacific countries to adopt this new and innovative technology.”
Mark Toland, president and chief executive officer of Corindus comments: “There is strong demand for robotic-assisted intervention from key opinion leaders and the broad clinical community in Japan. PMDA approval of CorPath GRX in Japan represents a significant step in our ongoing effort to bring cutting-edge vascular robotics to global markets. We believe robotics has the potential to enhance patient care in Japan by offering the benefits of robotic precision and advanced tools for physicians to perform these life-saving procedures.”











