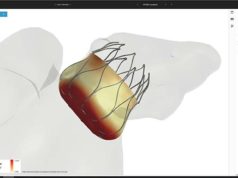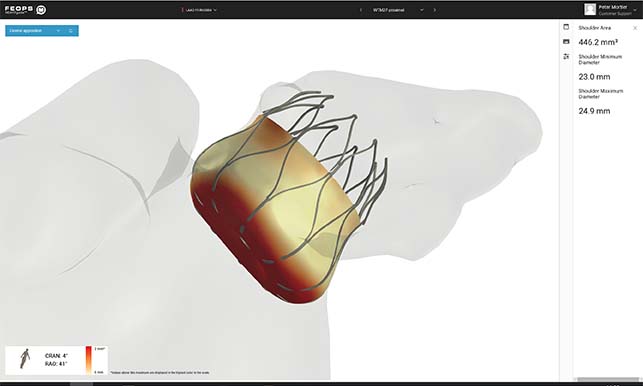
FEops has received CE mark approval for its FEops HEARTguide. According to a press release, FEops HEARTguide is a one-in-its-kind procedure planning environment for structural heart interventions that provides physicians with unique insights to evaluate device sizing and positioning preoperatively using novel computational modelling and simulation technology. The current release includes workflows for transcatheter aortic valve implantation (TAVI) and left atrial appendage (LAA) occlusion procedures.
The press release states that FEops HEARTguide helps to assess the risk of potential complications by preoperatively simulating multiple treatment scenarios in terms of device sizing and positioning, both for TAVI and LAA occlusion procedures. Within the TAVI workflow, it is also possible to plan TAVI procedures for patients with bicuspid aortic valve stenosis—the incidence of which is increasing with the shift towards treating low-risk patients.
Furthermore, the press release explains, FEops HEARTguide cloud-based procedure planning environment uses advanced personalised computational modelling and simulation to provide clinicians and medical device manufacturers with first-ever insights into the interaction between transcatheter structural heart devices and specific patient anatomy—preoperatively. The current release includes TAVI and LAA occlusion workflows. Such insights have the power to accelerate research and development of novel device-based solutions, as well as ultimately help to improve clinical outcomes in real-world hospital settings.
Cameron Dowling (St George’s Hospital, London, UK) states: “FEops HEARTguide gives our team increased confidence to perform TAVI in the most challenging of aortic valve anatomies”. Additionally, Ole De Backer (Rigshospitalet, Copenhagen, Denmark) notes: “I have the strong hope and believe that preprocedural planning with FEops HEARTguide will transform percutaneous LAA closure into an even more safe and more efficient procedure for patients with non-valvular atrial fibrillation and contraindications for oral anticoagulant therapy.”
Matthieu De Beule, CEO FEops, states: “I am extremely proud of the whole FEops team for reaching this important regulatory and commercial milestone and very pleased that we can offer our unique pre-operative planning support to the European clinical community. The European launch of FEops HEARTguide is a crucial next step to embody FEops’ vision to become the game-changer in the way transcatheter structural heart devices are designed and procedures are planned in the future.”