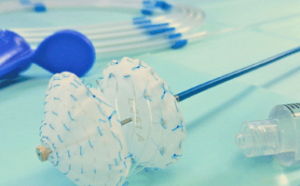
Carag has announced that it has completed CE marking for its Carag bioresorbable septal occluder. A press release reports that the device is a first-in-class structural heart device for treatment of common heart defects. It adds that the bioresorbable septal occluder was developed and clinically tested by Carag and is the world’s first transcatheter septal occluder, employing a framework that is fully resorbed by the human body.
Clinical testing of the device was performed by Horst Sievert (Cardiovascular Center, Frankfurt, Germany), where it was implanted in a series of patients with atrial septal defect or patent foramen ovale. The new implant demonstrated results equal to currently marketed metal framework (legacy) devices. Sievert says: “The Carag bioresorbable septal occluder is really a breakthrough! Since decades we have been waiting for a resorbable device for transcatheter closures. Patients have already heard about this resorbable device and are asking about its availability. I am looking forward to finally providing this alternative to my patients.”
Currently, atrial septal defect or patent foramen ovale are typically treated using catheter delivered implants composed of a metallic framework. Metal implants remain in the heart for the life of the patient, and are associated with major complications such as erosion, arrhythmia and thrombus formation.
Carag intends to perform a clinical trial in the USA under an Investigational Device Exemption (IDE), and is in negotiations with multiple international medical device companies for marketing rights.









