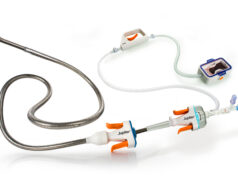
Biosensors International Group has announced both the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) approval for BioFreedom Ultra and US Food and Drug Administration (FDA) approval for BioFreedom.
BioFreedom is Biosensors’ novel polymer and carrier-free drug-coated stent with their proprietary limus drug Biolimus A9 (BA9). BA9 is Biosensors’ proprietary highly lipophilic anti-restenotic drug, developed specifically for use in coronary vascular applications. BioFreedom Ultra also features thin (84µmβ) Cobalt Chromium (Co/Cr) struts.
BioFreedom will give physicians in the USA the opportunity to reduce dual antiplatelet therapy (DAPT) to one month in patients post-PCI who are at High Risk of Bleeding (HBR).
Biosensors chief executive officer Yu Suhua, said: “We are excited to launch these coronary stent systems and broaden their availability in both Japan and the USA. The proven benefits of this Biolimus A9 coated stent system will help interventional cardiologists to further improve clinical outcomes for many patients requiring a stent, particularly those with a high bleeding risk.”
“Biosensors has been at the forefront of clinical stent research for the treatment of high-bleeding risk patients undergoing PCI [percutaneous coronary intervention] since the LEADERS FREE study was presented at TCT and published in the NEJM in 2015. It is great to finally be able to bring these technologies to Japan and the USA,” commented Biosensors chief medical officer Keith Oldroyd.
BioFreedom has become Biosensors’ first stent product to be approved in all the major key markets globally, as well as in many other country-specific registrations across the world, the company said in a press release.