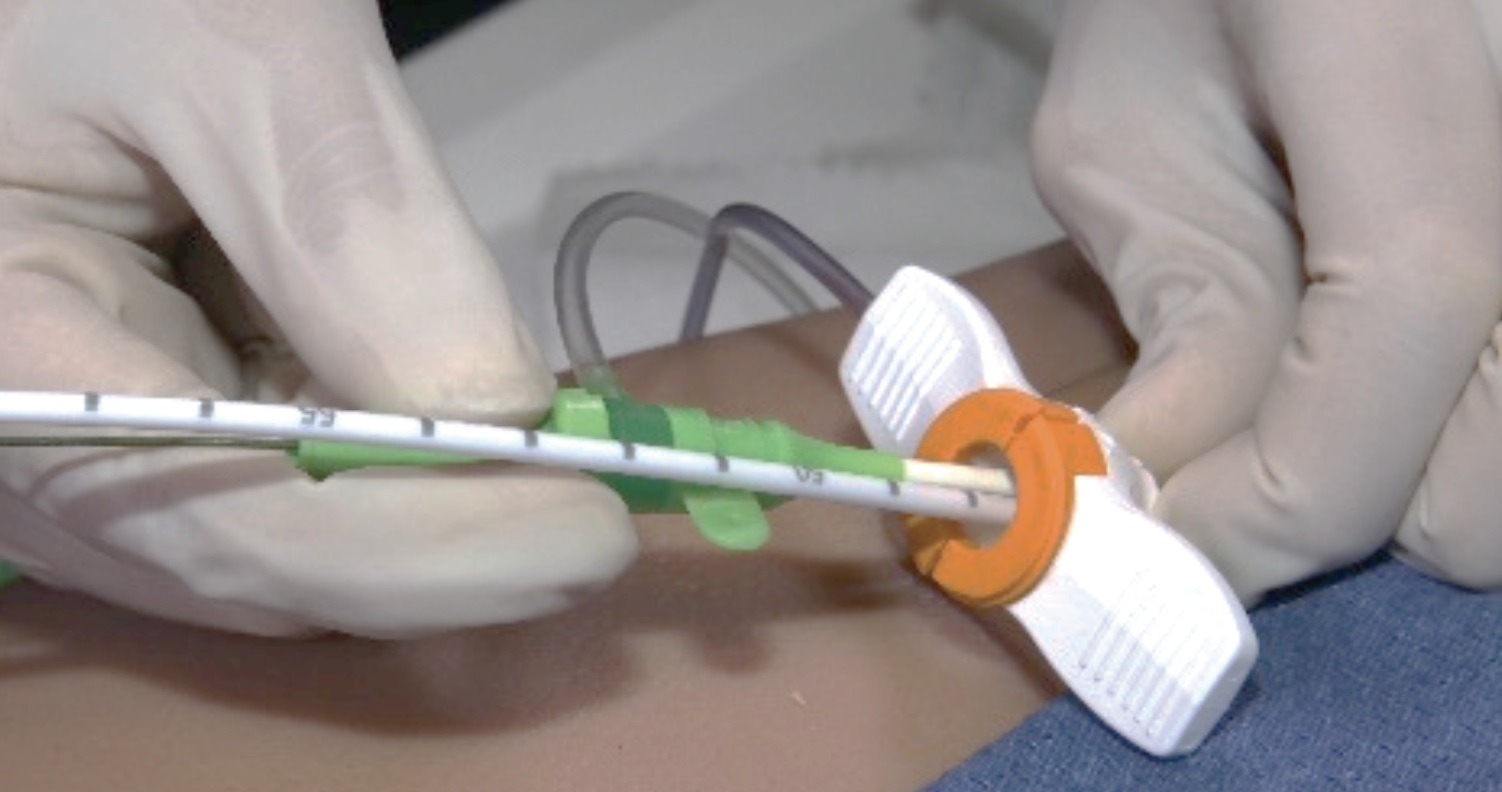
 Following consultation with its notified body, MEDCERT GmbH, Abiomed is introducing the single access procedure with Impella CP introducer sheaths for the treatment of heart disease in Europe.
Following consultation with its notified body, MEDCERT GmbH, Abiomed is introducing the single access procedure with Impella CP introducer sheaths for the treatment of heart disease in Europe.
Impella heart pumps have been developed to stabilise and recover the human heart, and the single access procedure enables the treating physician to place the device in the ventricle via a 14 French Impella CP introducer sheath, and to insert other catheters and introducer sheaths. Cardiac interventions such as percutaneous coronary intervention (PCI) can also be performed via the 14 French Impella CP introducer sheath.
The single access procedure is already practiced in the USA, where it was incorporated into Impella CP’s US labelling in January 2020. The confirmation given by MEDCERT GmbH for the single access procedure now gives cardiologists in Europe the option of performing interventional treatments for severe cardiac diseases via just one access site. This method may be beneficial for patients with limited vascular access and may reduce potential complications using only one access site.
MEDCERT GmbH is an organisation designated by Germany, as an EU member state, to determine whether medical devices comply with European directives and regulations. MEDCERT GmbH has now confirmed to Abiomed that the single access procedure is possible in Europe.










