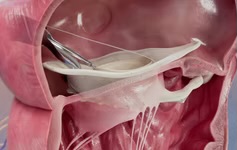
VDyne has announced that the US Food & Drug Administration (FDA) has approved an investigational device exemption (IDE) for the company’s pivotal clinical trial evaluating its transcatheter tricuspid valve replacement (TTVR) system—the TRIVITA1 IDE pivotal trial.
The IDE approval enables initiation of a US pivotal study at leading clinical centres to evaluate the safety and effectiveness of the VDyne system in patients with severe tricuspid regurgitation (TR).
“This is a defining milestone for VDyne and an important step toward bringing a much-needed therapy to patients with severe tricuspid regurgitation,” said Mike Buck, chief executive officer. “Our focus now is on disciplined clinical execution and partnering with leading investigators to generate high-quality data that advances the field and improves patient care.”
VDYNE’s TTVR system is designed as a patient-tailored, minimally invasive solution to address the complexities of tricuspid valve anatomy and disease. Since first-in-human use in November 2023, the system has been used globally through clinical studies and compassionate use, providing important insights into procedural performance and clinical outcomes.