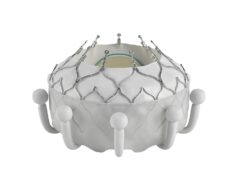
JenaValve has announced the completion of the 1,000th commercial procedure performed with its Trilogy transcatheter aortic valve implantation (TAVI) platform, performed in Cologne, Germany.
“This 1,000th case represents 1,000 patients who now have their lives back,” said Duane Pinto, chief medical officer at JenaValve. “This milestone speaks to the vast unmet need for patients suffering from aortic regurgitation (AR). We are quickly finding that there are indeed patients with AR that need less invasive solutions. In Europe, more and more patients are being referred that would not have historically been identified as a candidate for treatment or even diagnosed.”
“As an interventional cardiologist, I am constantly thinking about how to best treat patients whose options are limited,” said Matti Adam (University Hospital Cologne, Cologne, Germany). “Trilogy represents a fundamental shift in how we approach patients suffering from severe aortic regurgitation. These 1,000 implants represent patients who, just a few short years ago, would have had no viable options; yet today, after a procedure that I have found to be safe, reproducible, with consistent outcomes, they’re walking out of the hospital with their lives significantly improved.”
“A dedicated device like the Trilogy valve has given us an important new tool in our toolbox,” said Stephan Baldus (University Hospital Cologne, Cologne, Germany), who performed the case alongside Adam. “Oftentimes patients with aortic regurgitation are sent to our team quite late, largely due to lack of awareness for this disease. We previously would be faced with a challenging decision of performing open surgery on a sick patient, or attempting a TAVI with a device designed for aortic stenosis. The Trilogy has undoubtedly elevated outcomes for patients with AR.”
“We set out to transform the treatment landscape for severe AR and this milestone validates our mission,” said John Kilcoyne, chief executive officer of JenaValve. “This large and growing body of real-world evidence provides a strong foundation for future growth and adoption.”