Tag: CathWorks
Medtronic exercises its option to acquire CathWorks
Medtronic has announced it will exercise its option to acquire CathWorks.
The intent to acqui...
Enrolment completed in ALL-RISE trial of FFRangio coronary physiology assessment
CathWorks has announced the successful completion of enrolment of the Advancing Cath Lab Results...
Medtronic announces co-promotion agreement with CathWorks, with path toward acquisition
Medtronic today announced it has entered into a strategic partnership agreement with CathWorks, a pr...
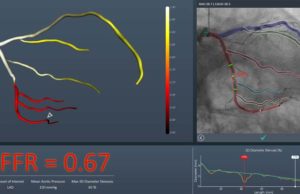
FFRangio system receives US FDA clearance
US FDA 510(k) clearance has been granted for the FFRangio System (CathWorks). The FFRangio syste...
TCT 2018: FFRangio may provide a faster and easier approach to physiological assessment
The FAST-FFR trial, which was presented at the 2018 Transcatheter Cardiovascular Therapeutics (T...
Enrolment in FAST-FFR trial completed early
CathWorks has announced that its FAST-FFR trial is fully enrolled ahead of schedule. The trial i...
CPT code for CathWorks’ 3D FFRangio wire free system
CathWorks has received a new CPT code 0523T for its non-invasive, 3D FFRangio system, which is d...