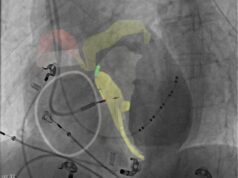
Protaryx Medical has submitted a 510(k) premarket notification to the US Food and Drug Administration (FDA) for its proprietary transseptal puncture device.
The device is a novel catheter system designed to improve the safety, accuracy and ease of transseptal puncture to facilitate access to the left atrium for procedures such as left atrial appendage closure (LAAC), transcatheter mitral valve procedures, and atrial fibrillation (AF) ablation. The device features an atraumatic, echogenic retractable nitinol mesh, as well as an extendable distal catheter, which are intended to enhance imaging visibility and aid deployment.
“Achieving 510(k) submission is a pivotal milestone for Protaryx,” said David Mester, chief executive officer of Protaryx. “Our team has worked with laser focus to solve one of the core workflow and ease-of-use challenges in structural heart procedures: transseptal puncture and left-atrial access. This submission brings us one step closer to offering a solution that enables physicians of all experience levels to operate with greater precision, less fluoroscopy, and a faster learning curve.”
Protaryx co-founder and system chief of cardiac surgery at Johns Hopkins Health System (Baltimore, USA), James Gammie, added: “The results from the FIH [first-in-human] study show that this device not only meets unmet needs in transseptal access but has the potential to set a new standard for safety, ease-of-use, and efficiency in left-heart interventions.”