Meril Life Sciences has announced the release of one-year outcomes from the pivotal LANDMARK randomiced controlled trial (RCT) during a late-breaking trials session at PCR London Valves 2025 (16–18 November, London, UK).
The LANDMARK trial is a direct comparison of the balloon-expandable Myval transcatheter heart valve (THV) series with both the balloon-expandable Sapien (Edwards Lifesciences) and the self-expanding Evolut Medtronic in patients with symptomatic severe aortic stenosis.
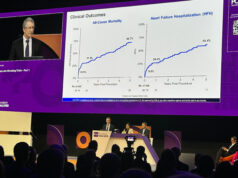
At one-year, the Myval THV series demonstrated comparable composite clinical efficacy, defined as freedom from all-cause mortality, all stroke, and procedure- or valve-related hospitalisation, when compared to the Sapien and Evolut THV devices, with outcomes reported at 87% for Myval, 86.9% for Sapien, and 86.9% for Evolut.
The trial also assessed an extended clinical efficacy endpoint (freedom from all-cause mortality, all stroke, procedure- or valve-related hospitalisation and quality of life deterioration) reflecting sustained clinical benefit over time. These extended outcomes remained consistent across platforms, measured at 80.5% for Myval, 75% for Sapien, and 79.7% for Evolut. The Myval THV series demonstrated the lowest incidence of moderate aortic regurgitation, recorded at 1.6%, the company said in a press release.
A post-hoc analysis in patients with small aortic annulus confirmed comparable one-year composite efficacy outcomes (freedom from all-cause mortality, all stroke, and procedure- or valve-related hospitalisation), with results of 91% for Myval versus 89% for Sapien, and 91% for Evolut.
“The LANDMARK trial continues to provide the global TAVI community with meaningful comparative insight,” LANDMARK study chairman and director Patrick W Serruys (National University of Ireland, Galway, Ireland). “By bringing together three leading THV platforms in a rigorously designed randomised study, we are able to better understand differences that matter clinically, particularly in valve stability and sustained haemodynamic performance. The one-year findings reinforce the reliability of the Myval THV series, including its low rate of aortic regurgitation, which is of significant relevance for valve performance. These data support informed clinical decision-making and expand the conversation on how to personalize valve selection according to patient anatomy.”
“This study is unique in its ability to benchmark performance across balloon-expandable and self-expanding valves under identical trial conditions,” said Andreas Baumbach (Queen Mary University of London, London, UK), LANDMARK global principal investigator. “The consistency seen in both standard and extended clinical efficacy endpoints demonstrates that the Myval THV series performs on par with established global systems. Its haemodynamic profile, combined with predictable deployment and sizing options, provides clinicians with a valuable and versatile treatment choice. As the study progresses over the coming years, it will further contribute to greater clarity on long-term valve performance, which is increasingly important given the demographic shift toward younger and lower-risk TAVI patients.”
“The LANDMARK trial reflects our commitment to building robust clinical evidence that enables confident clinical decision making,” Sanjeev Bhatt, senior vice president–corporate strategy at Meril, said: “The continued performance of the Myval THV series across diverse patient populations, including those with complex or smaller anatomies, reinforces its role as a next-generation therapy solution. Our focus at Meril is to develop globally relevant, scientifically driven innovations that expand access to advanced structural heart care. These results further strengthen the foundation for Myval’s continued adoption across different geographies and clinical practice environments.”
The LANDMARK trial will continue patient follow-up for a period of ten years to evaluate valve performance, long-term clinical stability, and echocardiographic performance.