The first-in-man use of the ventriculo-coronary transcatheter outward navigation and reentry—VECTOR—technique, a closed-chest, transcatheter, coronary ostial bypass procedure, has been reported in Circulation: Cardiovascular Interventions.
Adam Greenbaum (Emory School of Medicine, Atlanta, USA) and colleagues from the US National Institutes of Health (NIH) detail the case involving a 67-year-old male patient treated for bioprosthetic aortic valve degeneration. Investigators used the VECTOR procedure to bypass the patient’s left main stem prior to transcatheter aortic valve implantation (TAVI), relocating the coronary ostium to avoid obstruction of the coronary artery by the transcatheter valve.
The procedure uses covered stents and catheter techniques resembling retrograde chronic total occlusion revascularisation and may offer a less traumatic alternative to open-heart surgery for those at risk of coronary artery obstruction.
In their paper, Greenbaum et al detail that the patient had undergone previous aortic and mitral valve replacements and had developed decompensated heart failure due to severe stenotic degradation of the aortic bioprosthesis. Comorbidities included end-stage renal failure requiring haemodialysis, non-disabling stroke, a severe non-ischaemic cardiomyopathy, and severe peripheral arterial disease. After heart team discussion, it was decided that the patient was at prohibitive risk for redo surgery due to the presence of advanced comorbidities and low rehabilitation potential.
“Our patient had an extensive history of prior interventions, vascular disease, and other confounders, which meant that open-heart surgery was completely off the table. Having a minimally invasive alternative in a case like this is paramount,” comments Greenbaum in a press release issued by NIH following the publication of the case report detailing the procedure.
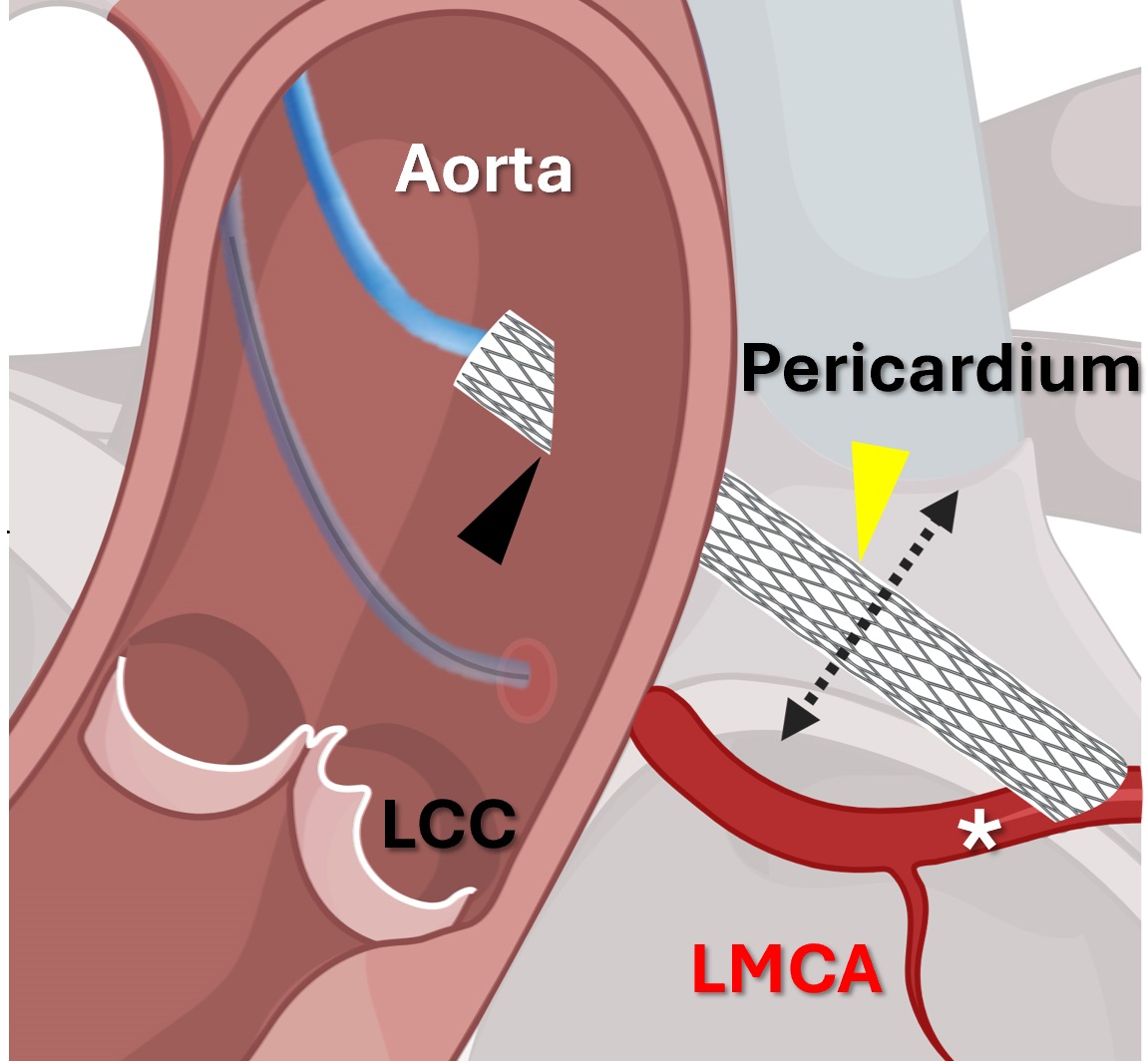
In their Circulation: Cardiovascular Interventions paper, the researchers detail how the patient’s unique anatomy limited the options available to them. Cardiac computed tomography (CT) showed that the bioprosthetic valve frame extended above both the left coronary artery and sinotubular junction, indicating extreme risk for left coronary artery obstruction.
“We thought, ‘why don’t we just move the ostium of the coronary artery out of the danger zone?’,” Greenbaum is quoted as saying in the press release.
The procedure creates a new route for blood flow that is a safe distance away from the aortic valve using the vascular circuitry to reach the heart without the need for a thoracotomy. A wire is passed through the aorta and into the at-risk coronary artery. From there, the wire is steered deep into one of the artery’s branches, breaching the vessel into the right ventricle. There, a separate catheter is operated to ensnare that wire and then pull the wire’s end out through the femoral vein. Now a continuous line from aorta to vein, this wire enables loading of more sophisticated tools into the target artery.
The next goal of VECTOR is to produce a new ostium for the coronary bypass. The operators create one hole in the aorta downstream from the valve, out of range of potential blockage. They then make a second opening by piercing through the coronary artery wall using a special catheter, which is braced by an expandable stent. They pass two loose ends through each of the holes and then, as in the previous phase, tie them together to create another bridge, this time tracing a safe path for the bypass.
Using this second wire, the team feeds a coronary bypass graft through the two new openings. Once deployed, the graft provides a new route for blood flow.
Six months after the procedure, the patient showed no signs of coronary artery obstruction, meaning that the first use of the procedure in a human proved to be a success, the researchers have stated.
The authors suggest the new technique may also find some footing in treating coronary diseases more broadly, such as in calcified ostial coronary lesions or recurrent in-stent restenosis.
“The technique was accomplished in a hostile, postsurgical, pericardium with extensive adhesions. Longevity of the covered stent conduit remains uncertain; however, we hypothesise, based on experience with extra-anatomic venous conduits, that the extra-vascular course may prevent restenosis,” Greenbaum and colleagues write in their case report in Circulation: Cardiovascular Interventions. “Further procedures, in highly selected patients, are required to demonstrate reproducibility, shorten procedure time, and determine if the use of extracorporeal circulatory support is uniformly necessary.”