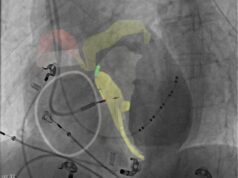
Nyra Medical has announced the initiation of its first-in-human clinical study evaluating its novel Cardiac Leaflet Enhancer (CARLEN) system in patients with functional (secondary) mitral regurgitation (FMR).
The study, focused on clinical safety and technical performance, has enrolled more than 10 patients, many with at least thirty-day follow-up to date.
Nyra Medical’s proprietary transcatheter technology is designed to repair FMR by augmenting the native mitral valve. The CARLEN system is a single-leaflet implant designed to restore coaptation while preserving the valve’s natural motion and orifice area.
With the ability to implant on either the anterior or posterior leaflets, the CARLEN system offers broad clinical applicability and minimal anatomical or clinical exclusions, Nyra Medical says in a press release. The design aims to address the anatomical and functional challenges that have limited the success of earlier therapies, the press release states.
“Achieving first-in-human data is a major milestone event for any medtech company,” said Lori Chmura, chief executive officer of Nyra Medical. “Nyra’s early clinical results underscore the potential of this platform to redefine how we treat structural heart disease.”
Nyra Medical says that its solution intends to offer a differentiated, minimally invasive approach with broad potential applicability.
“We are entering a new era of transcatheter therapy where meaningful mitral repair is finally within reach. The data we’ve seen so far is compelling and gives us confidence to push forward thoughtfully but decisively,” said Azeem Latib (Montefiore Medical Center, New York, USA).
Susheel Kodali (New York Presbyterian Medical Center, New York, USA) added: “This approach offers a promising path forward for patients. The early results suggest we may be on the cusp of an important clinical advancement.”
The CARLEN implantation procedure leverages many parallels to the established workflow, utilising familiar echocardiographic guidance and intraprocedural imaging to support precise positioning and real-time assessment of valve function.
Edwin Ho (Montefiore Medical Center, New York, USA), commented: “For interventional echocardiographers, CARLEN introduces innovation without adding complexity. By maintaining the familiar imaging workflow, we can focus on optimising outcomes with confidence as we visualise leaflet engagement and function with remarkable clarity.”
The ongoing first-in-human study is being conducted at international centers in the Republic of Georgia, Paraguay and Uzbekistan with enrolment expected to broaden over the coming months as additional patients are screened and treated. Insights gained from the early clinical experience will play a critical role in refining procedural workflows and device performance ahead of expanded clinical investigation.
The data will directly inform the company’s global regulatory strategy, including engagement with the US Food and Drug Administration (FDA) and other regulatory bodies. The findings will shape the design of future clinical studies, including a potential early feasibility study and pivotal trial in the USA. Early analysis from the first-in-human study will be presented at the upcoming TransCatheter Therapeutics (TCT) conference (25–28 October, San Francisco, USA) by Kodali.