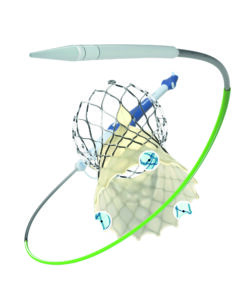
 Medtronic has received US Food and Drug Administration (FDA) approval for the expanded redo-transcatheter aortic valve implantation (TAVI) indication of the Evolut TAVI system.
Medtronic has received US Food and Drug Administration (FDA) approval for the expanded redo-transcatheter aortic valve implantation (TAVI) indication of the Evolut TAVI system.
This approval allows for the implantation of a new Evolut transcatheter aortic valve (TAV) inside any failed previously implanted transcatheter aortic valve (TAV) and follows CE mark for Evolut PRO+, FX and FX+ systems.
The Redo-TAVI procedure is indicated for patients experiencing failure of any TAV, including but not limited to severe aortic stenosis, who are at high-risk for open-heart surgery.
“FDA approval for Redo-TAVI with the Evolut system marks a significant milestone in patient care, empowering physicians across the USA to offer a critical treatment option for patients with failing transcatheter heart valves who are at high surgical risk,” said Michael Caskey, attending cardiothoracic surgeon at Abrazo Arizona Heart Hospital in Phoenix (USA). “This advancement also benefits patients considering a new TAVI procedure today, giving patients options for future intervention and has the potential to profoundly improve their long-term outcomes and quality of life.”
“The Redo-TAVI indication marks an important milestone for our Evolut TAVI systems, reinforcing our commitment to provide physicians with patient-specific solutions today, and for the future of TAVI,” said Jorie Soskin, vice president and general manager of the Structural Heart business within the Cardiovascular Portfolio at Medtronic. “With a broader indication than other available options, this expansion, together with the launch of our RESTORE study build on our differentiated design and unparalleled evidence to advance solutions and elevate care options for heart teams and patients worldwide.”
In addition to the FDA approval, Medtronic has launched the RESTORE study to evaluate the outcomes of Redo TAVI in patients experiencing symptomatic bioprosthetic valve failure. This pivotal study will enrol 225 participants and follow them for up to five years, focusing on both immediate and long-term clinical outcomes. The study aims to assess 30-day procedural success rates, one-year freedom from mortality and stroke, and additional measures of safety, technical success, and quality of life.
In native aortic valves, Evolut TAVI systems are indicated for implantation in symptomatic severe aortic stenosis patients across all risk categories (extreme, high, intermediate, and low) in more than 120 countries in the world, including the European Union and the USA.